Heterologous expression of moss light-harvesting complex stress-related 1 (LHCSR1), the chlorophyll a-xanthophyll pigment-protein complex catalyzing non-photochemical quenching, in Nicotiana sp
- PMID: 26260788
- PMCID: PMC4591818
- DOI: 10.1074/jbc.M115.668798
Heterologous expression of moss light-harvesting complex stress-related 1 (LHCSR1), the chlorophyll a-xanthophyll pigment-protein complex catalyzing non-photochemical quenching, in Nicotiana sp
Abstract
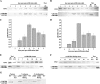
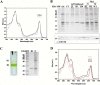
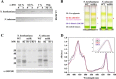
Oxygenic photosynthetic organisms evolved mechanisms for thermal dissipation of energy absorbed in excess to prevent formation of reactive oxygen species. The major and fastest component, called non-photochemical quenching, occurs within the photosystem II antenna system by the action of two essential light-harvesting complex (LHC)-like proteins, photosystem II subunit S (PSBS) in plants and light-harvesting complex stress-related (LHCSR) in green algae and diatoms. In the evolutionary intermediate Physcomitrella patens, a moss, both gene products are active. These proteins, which are present in low amounts, are difficult to purify, preventing structural and functional analysis. Here, we report on the overexpression of the LHCSR1 protein from P. patens in the heterologous systems Nicotiana benthamiana and Nicotiana tabacum using transient and stable nuclear transformation. We show that the protein accumulated in both heterologous systems is in its mature form, localizes in the chloroplast thylakoid membranes, and is correctly folded with chlorophyll a and xanthophylls but without chlorophyll b, an essential chromophore for plants and algal LHC proteins. Finally, we show that recombinant LHCSR1 is active in quenching in vivo, implying that the recombinant protein obtained is a good material for future structural and functional studies.
Keywords: LHCSR; NPQ; Physcomitrella; light-harvesting complex (antenna complex); photobiology; photosynthesis; photosynthetic pigment; recombinant protein expression; tobacco.
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures








Similar articles
-
Functional analysis of LHCSR1, a protein catalyzing NPQ in mosses, by heterologous expression in Arabidopsis thaliana.Photosynth Res. 2019 Dec;142(3):249-264. doi: 10.1007/s11120-019-00656-3. Epub 2019 Jul 3. Photosynth Res. 2019. PMID: 31270669 Free PMC article.
-
The rise and fall of Light-Harvesting Complex Stress-Related proteins as photoprotection agents during evolution.J Exp Bot. 2019 Oct 24;70(20):5527-5535. doi: 10.1093/jxb/erz317. J Exp Bot. 2019. PMID: 31424076
-
The evolution of the photoprotective antenna proteins in oxygenic photosynthetic eukaryotes.Biochem Soc Trans. 2018 Oct 19;46(5):1263-1277. doi: 10.1042/BST20170304. Epub 2018 Aug 28. Biochem Soc Trans. 2018. PMID: 30154089 Review.
-
Electron transfer between carotenoid and chlorophyll contributes to quenching in the LHCSR1 protein from Physcomitrella patens.Biochim Biophys Acta. 2016 Dec;1857(12):1870-1878. doi: 10.1016/j.bbabio.2016.09.001. Epub 2016 Sep 7. Biochim Biophys Acta. 2016. PMID: 27614061
-
Energy-Dependent Non-Photochemical Quenching: PsbS, LhcSR, and Other Players.Biochemistry (Mosc). 2025 Jan;90(1):44-60. doi: 10.1134/S000629792460371X. Biochemistry (Mosc). 2025. PMID: 40058973 Review.
Cited by
-
Genetic Engineering, Synthetic Biology and the Light Reactions of Photosynthesis.Plant Physiol. 2019 Mar;179(3):778-793. doi: 10.1104/pp.18.00360. Epub 2018 Jul 10. Plant Physiol. 2019. PMID: 29991483 Free PMC article. Review.
-
Single-molecule spectroscopy of LHCSR1 protein dynamics identifies two distinct states responsible for multi-timescale photosynthetic photoprotection.Nat Chem. 2017 Aug;9(8):772-778. doi: 10.1038/nchem.2818. Epub 2017 Jul 17. Nat Chem. 2017. PMID: 28754946
-
Microsecond and millisecond dynamics in the photosynthetic protein LHCSR1 observed by single-molecule correlation spectroscopy.Proc Natl Acad Sci U S A. 2019 Jun 4;116(23):11247-11252. doi: 10.1073/pnas.1821207116. Epub 2019 May 17. Proc Natl Acad Sci U S A. 2019. PMID: 31101718 Free PMC article.
-
Recent advances in expression and purification strategies for plant made vaccines.Front Plant Sci. 2023 Nov 23;14:1273958. doi: 10.3389/fpls.2023.1273958. eCollection 2023. Front Plant Sci. 2023. PMID: 38078091 Free PMC article. Review.
-
Functional analysis of LHCSR1, a protein catalyzing NPQ in mosses, by heterologous expression in Arabidopsis thaliana.Photosynth Res. 2019 Dec;142(3):249-264. doi: 10.1007/s11120-019-00656-3. Epub 2019 Jul 3. Photosynth Res. 2019. PMID: 31270669 Free PMC article.
References
-
- Barber J., Andersson B. (1992) Too much of a good thing: light can be bad for photosynthesis. Trends Biochem. Sci. 17, 61–66 - PubMed
-
- Miller G., Shulaev V., Mittler R. (2008) Reactive oxygen signaling and abiotic stress. Physiol. Plant. 133, 481–489 - PubMed
-
- Takahashi S., Murata N. (2008) How do environmental stresses accelerate photoinhibition? Trends Plant Sci. 13, 178–182 - PubMed
-
- Genty B., Briantais J. M., Baker N. R. (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta 990, 87–92
-
- Niyogi K. K. (2000) Safety valves for photosynthesis. Curr. Opin. Plant Biol. 3, 455–460 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

