A Bayesian adaptive design for biomarker trials with linked treatments
- PMID: 26263479
- PMCID: PMC4559835
- DOI: 10.1038/bjc.2015.278
A Bayesian adaptive design for biomarker trials with linked treatments
Abstract
Background: Response to treatments is highly heterogeneous in cancer. Increased availability of biomarkers and targeted treatments has led to the need for trial designs that efficiently test new treatments in biomarker-stratified patient subgroups.
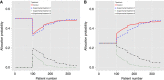
Methods: We propose a novel Bayesian adaptive randomisation (BAR) design for use in multi-arm phase II trials where biomarkers exist that are potentially predictive of a linked treatment's effect. The design is motivated in part by two phase II trials that are currently in development. The design starts by randomising patients to the control treatment or to experimental treatments that the biomarker profile suggests should be active. At interim analyses, data from treated patients are used to update the allocation probabilities. If the linked treatments are effective, the allocation remains high; if ineffective, the allocation changes over the course of the trial to unlinked treatments that are more effective.
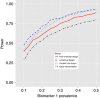
Results: Our proposed design has high power to detect treatment effects if the pairings of treatment with biomarker are correct, but also performs well when alternative pairings are true. The design is consistently more powerful than parallel-groups stratified trials.
Conclusions: This BAR design is a powerful approach to use when there are pairings of biomarkers with treatments available for testing simultaneously.
Figures

References
-
- Adams CP, Brantner VV. Estimating the cost of new drug development: is it really $802 million. Health Aff. 2006;25 (2:420–428. - PubMed
-
- Barker AD, Sigman CC, Kelloff GJ, Hylton NM, Berry DA, Esserman LJ. I-SPY 2: an adaptive breast cancer trial design in the setting of neoadjuvant chemotherapy. Clin Pharmacol Ther. 2009;86 (1:97–100. - PubMed
-
- Berry SM, Carlin BP, Lee JJ, Muller P. Bayesian adaptive methods for clinical trials. CRC press; 2010.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

