Glucose intolerance induced by blockade of central FGF receptors is linked to an acute stress response
- PMID: 26266088
- PMCID: PMC4529498
- DOI: 10.1016/j.molmet.2015.05.005
Glucose intolerance induced by blockade of central FGF receptors is linked to an acute stress response
Abstract
Objective: Central administration of ligands for fibroblast growth factor receptors (FGFRs) such as fibroblast growth factor-19 (FGF19) and FGF21 exert glucose-lowering effects in rodent models of obesity and type 2 diabetes (T2D). Conversely, intracerebroventricular (icv) administration of the non-selective FGFR inhibitor (FGFRi) PD173074 causes glucose intolerance, implying a physiological role for neuronal FGFR signaling in glucose homeostasis. The current studies were undertaken to identify neuroendocrine mechanisms underlying the glucose intolerance induced by pharmacological blockade of central FGFRs.
Methods: Overnight fasted, lean, male, Long-Evans rats received icv injections of either PD173074 or vehicle (Veh) followed 30 min later by performance of a frequently sampled intravenous glucose tolerance test (FSIGT). Minimal model analysis of glucose and insulin data from the FSIGT was performed to estimate insulin-dependent and insulin-independent components of glucose disposal. Plasma levels of lactate, glucagon, corticosterone, non-esterified free fatty acids (NEFA) and catecholamines were measured before and after intravenous (iv) glucose injection.
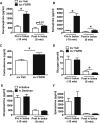
Results: Within 20 min of icv PD173074 injection (prior to the FSIGT), plasma levels of lactate, norepinephrine and epinephrine increased markedly, and each returned to baseline rapidly (within 8 min) following the iv glucose bolus. In contrast, plasma glucagon levels were not altered by icv FGFRi at either time point. Consistent with a previous report, glucose tolerance was impaired following icv PD173074 compared to Veh injection and, based on minimal model analysis of FSIGT data, this effect was attributable to reductions of both insulin secretion and the basal insulin effect (BIE), consistent with the inhibitory effect of catecholamines on pancreatic β-cell secretion. By comparison, there were no changes in glucose effectiveness at zero insulin (GEZI) or the insulin sensitivity index (SI). To determine if iv glucose (given during the FSIGT) contributed to the rapid resolution of the sympathoadrenal response induced by icv FGFRi, we performed an additional study comparing groups that received iv saline or iv glucose 30 min after icv FGFRi. Our finding that elevated plasma catecholamine levels returned rapidly to baseline irrespective of whether rats subsequently received an iv bolus of saline or glucose indicates that the rapid reversal of sympathoadrenal activation following icv FGFRi was unrelated to the subsequent glucose bolus.
Conclusions: The effect of acute inhibition of central FGFR signaling to impair glucose tolerance likely involves a stress response associated with pronounced, but transient, sympathoadrenal activation and an associated reduction of insulin secretion. Whether this effect is a true consequence of FGFR blockade or involves an off-target effect of the FGFR inhibitor requires additional study.
Keywords: AIRg, acute insulin response to glucose; AUC, area under the curve; BIE, basal insulin effect; CNS, central nervous system; Central FGF receptors; DI, disposition index; FGF receptor inhibitor PD173074; FGF, fibroblast growth factor; FGF19, Fibroblast growth factor-19; FGFR, fibroblast growth factor receptor; FGFRi, FGFR inhibitor; FSIGT, frequently sampled intravenous glucose tolerance test; Frequently sampled intravenous glucose tolerance test; GE, glucose effectiveness; GEZI, glucose effectiveness at zero insulin; Glucose metabolism; Ibasal, basal insulin; Kg, glucose disappearance constant; Minimal model; NEFA, non-esterified free fatty acids; SG, glucose effectiveness at basal insulin; SI, insulin sensitivity; Sympathoadrenal response; icv, intracerebroventricular.
Figures


References
-
- Fu L., John L.M., Adams S.H., Yu X.X., Tomlinson E., Renz M. Fibroblast growth factor 19 increases metabolic rate and reverses dietary and leptin-deficient diabetes. Endocrinology. 2004;145:2594–2603. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

