Expression of the Acidothermus cellulolyticus E1 endoglucanase in Caldicellulosiruptor bescii enhances its ability to deconstruct crystalline cellulose
- PMID: 26269712
- PMCID: PMC4533959
- DOI: 10.1186/s13068-015-0296-x
Expression of the Acidothermus cellulolyticus E1 endoglucanase in Caldicellulosiruptor bescii enhances its ability to deconstruct crystalline cellulose
Abstract
Background: The Caldicellulosiruptor bescii genome encodes a potent set of carbohydrate-active enzymes (CAZymes), found primarily as multi-domain enzymes that exhibit high cellulolytic and hemicellulolytic activity on and allow utilization of a broad range of substrates, including plant biomass without conventional pretreatment. CelA, the most abundant cellulase in the C. bescii secretome, uniquely combines a GH9 endoglucanase and a GH48 exoglucanase in one protein. The most effective commercial enzyme cocktails used in vitro to pretreat biomass are derived from fungal cellulases (cellobiohydrolases, endoglucanases and a β-d-glucosidases) that act synergistically to release sugars for microbial conversion. The C. bescii genome contains six GH5 domains in five different open reading frames. Four exist in multi-domain proteins and two as single catalytic domains. E1 is a GH5 endoglucanase reported to have high specific activity and simple architecture and is active at the growth temperature of C. bescii. E1 is an endo-1,4-β-glucanase linked to a family 2 carbohydrate-binding module shown to bind primarily to cellulosic substrates. We tested if the addition of this protein to the C. bescii secretome would improve its cellulolytic activity.
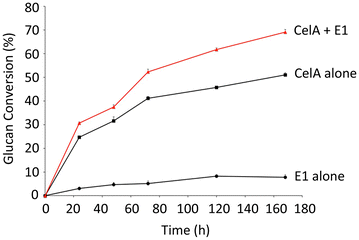
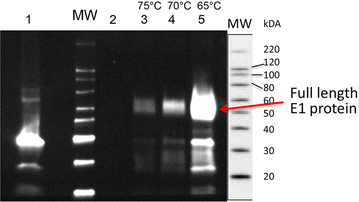
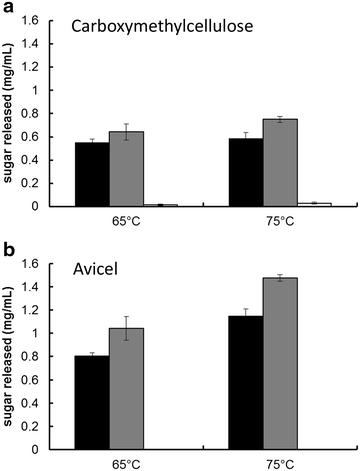
Results: In vitro analysis of E1 and CelA shows synergistic interaction. The E1 gene from Acidothermus cellulolyticus was cloned and expressed in C. bescii under the transcriptional control of the C. bescii S-layer promoter, and secretion was directed by the addition of the C. bescii CelA signal peptide sequence. The vector was integrated into the C. bescii chromosome at a site previously showing no detectable detrimental consequence. Increased activity of the secretome of the strain containing E1 was observed on both carboxymethylcellulose (CMC) and Avicel. Activity against CMC increased on average 10.8 % at 65 °C and 12.6 % at 75 °C. Activity against Avicel increased on average 17.5 % at 65 °C and 16.4 % at 75 °C.
Conclusions: Expression and secretion of E1 in C. bescii enhanced the cellulolytic ability of its secretome. These data agree with in vitro evidence that E1 acts synergistically with CelA to digest cellulose and offer the possibility of engineering additional enzymes for improved biomass deconstruction with the knowledge that C. bescii can express a gene from Acidothermus, and perhaps other heterologous genes, effectively.
Figures

Similar articles
-
Heterologous co-expression of two β-glucanases and a cellobiose phosphorylase resulted in a significant increase in the cellulolytic activity of the Caldicellulosiruptor bescii exoproteome.J Ind Microbiol Biotechnol. 2019 May;46(5):687-695. doi: 10.1007/s10295-019-02150-0. Epub 2019 Feb 20. J Ind Microbiol Biotechnol. 2019. PMID: 30783893
-
Deletion of Caldicellulosiruptor bescii CelA reveals its crucial role in the deconstruction of lignocellulosic biomass.Biotechnol Biofuels. 2014 Oct 9;7(1):142. doi: 10.1186/s13068-014-0142-6. eCollection 2014. Biotechnol Biofuels. 2014. PMID: 25317205 Free PMC article.
-
Functional Analysis of the Glucan Degradation Locus in Caldicellulosiruptor bescii Reveals Essential Roles of Component Glycoside Hydrolases in Plant Biomass Deconstruction.Appl Environ Microbiol. 2017 Dec 1;83(24):e01828-17. doi: 10.1128/AEM.01828-17. Print 2017 Dec 15. Appl Environ Microbiol. 2017. PMID: 28986379 Free PMC article.
-
Engineering the N-terminal end of CelA results in improved performance and growth of Caldicellulosiruptor bescii on crystalline cellulose.Biotechnol Bioeng. 2017 May;114(5):945-950. doi: 10.1002/bit.26242. Biotechnol Bioeng. 2017. PMID: 28019666
-
Metabolic engineering of Caldicellulosiruptor bescii for hydrogen production.Appl Microbiol Biotechnol. 2024 Dec;108(1):65. doi: 10.1007/s00253-023-12974-7. Epub 2024 Jan 9. Appl Microbiol Biotechnol. 2024. PMID: 38194138 Free PMC article. Review.
Cited by
-
Coexpression of a β-d-Xylosidase from Thermotoga maritima and a Family 10 Xylanase from Acidothermus cellulolyticus Significantly Improves the Xylan Degradation Activity of the Caldicellulosiruptor bescii Exoproteome.Appl Environ Microbiol. 2021 Jun 25;87(14):e0052421. doi: 10.1128/AEM.00524-21. Epub 2021 Jun 25. Appl Environ Microbiol. 2021. PMID: 33990300 Free PMC article.
-
Heterologous expression of a β-D-glucosidase in Caldicellulosiruptor bescii has a surprisingly modest effect on the activity of the exoproteome and growth on crystalline cellulose.J Ind Microbiol Biotechnol. 2017 Dec;44(12):1643-1651. doi: 10.1007/s10295-017-1982-4. Epub 2017 Sep 23. J Ind Microbiol Biotechnol. 2017. PMID: 28942503
-
High activity CAZyme cassette for improving biomass degradation in thermophiles.Biotechnol Biofuels. 2018 Feb 1;11:22. doi: 10.1186/s13068-018-1014-2. eCollection 2018. Biotechnol Biofuels. 2018. PMID: 29434665 Free PMC article.
-
Cloning, Expression, and Characterization of a Thermophilic Endoglucanase, AcCel12B from Acidothermus cellulolyticus 11B.Int J Mol Sci. 2015 Oct 22;16(10):25080-95. doi: 10.3390/ijms161025080. Int J Mol Sci. 2015. PMID: 26506341 Free PMC article.
-
Native xylose-inducible promoter expands the genetic tools for the biomass-degrading, extremely thermophilic bacterium Caldicellulosiruptor bescii.Extremophiles. 2018 Jul;22(4):629-638. doi: 10.1007/s00792-018-1023-x. Epub 2018 May 24. Extremophiles. 2018. PMID: 29797090
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

