APP intracellular domain-WAVE1 pathway reduces amyloid-β production
- PMID: 26280122
- PMCID: PMC4560977
- DOI: 10.1038/nm.3924
APP intracellular domain-WAVE1 pathway reduces amyloid-β production
Abstract
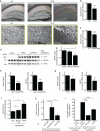
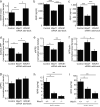
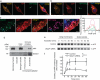
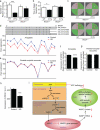
An increase in amyloid-β (Aβ) production is a major pathogenic mechanism associated with Alzheimer's disease (AD), but little is known about possible homeostatic control of the amyloidogenic pathway. Here we report that the amyloid precursor protein (APP) intracellular domain (AICD) downregulates Wiskott-Aldrich syndrome protein (WASP)-family verprolin homologous protein 1 (WAVE1 or WASF1) as part of a negative feedback mechanism to limit Aβ production. The AICD binds to the Wasf1 promoter, negatively regulates its transcription and downregulates Wasf1 mRNA and protein expression in Neuro 2a (N2a) cells. WAVE1 interacts and colocalizes with APP in the Golgi apparatus. Experimentally reducing WAVE1 in N2a cells decreased the budding of APP-containing vesicles and reduced cell-surface APP, thereby reducing the production of Aβ. WAVE1 downregulation was observed in mouse models of AD. Reduction of Wasf1 gene expression dramatically reduced Aβ levels and restored memory deficits in a mouse model of AD. A decrease in amounts of WASF1 mRNA was also observed in human AD brains, suggesting clinical relevance of the negative feedback circuit involved in homeostatic regulation of Aβ production.
Figures
Similar articles
-
Huntingtin associated protein 1 regulates trafficking of the amyloid precursor protein and modulates amyloid beta levels in neurons.J Neurochem. 2012 Sep;122(5):1010-22. doi: 10.1111/j.1471-4159.2012.07845.x. J Neurochem. 2012. PMID: 22731248
-
Amyloid-β protein (Aβ) Glu11 is the major β-secretase site of β-site amyloid-β precursor protein-cleaving enzyme 1(BACE1), and shifting the cleavage site to Aβ Asp1 contributes to Alzheimer pathogenesis.Eur J Neurosci. 2013 Jun;37(12):1962-9. doi: 10.1111/ejn.12235. Eur J Neurosci. 2013. PMID: 23773065
-
Presenilin 1 regulates the processing of beta-amyloid precursor protein C-terminal fragments and the generation of amyloid beta-protein in endoplasmic reticulum and Golgi.Biochemistry. 1998 Nov 24;37(47):16465-71. doi: 10.1021/bi9816195. Biochemistry. 1998. PMID: 9843412
-
From synaptic spines to nuclear signaling: nuclear and synaptic actions of the amyloid precursor protein.J Neurochem. 2013 Jul;126(2):183-90. doi: 10.1111/jnc.12239. Epub 2013 Apr 3. J Neurochem. 2013. PMID: 23495999 Review.
-
Possible roles of amyloid intracellular domain of amyloid precursor protein.BMB Rep. 2010 Oct;43(10):656-63. doi: 10.5483/BMBRep.2010.43.10.656. BMB Rep. 2010. PMID: 21034527 Review.
Cited by
-
Systematic Analysis of Biological Processes Reveals Gene Co-expression Modules Driving Pathway Dysregulation in Alzheimer's Disease.Aging Dis. 2024 Jun 12;16(3):1598-1625. doi: 10.14336/AD.2024.0429. Aging Dis. 2024. PMID: 38913039 Free PMC article.
-
Regulatory feedback cycle of the insulin-degrading enzyme and the amyloid precursor protein intracellular domain: Implications for Alzheimer's disease.Aging Cell. 2020 Nov;19(11):e13264. doi: 10.1111/acel.13264. Epub 2020 Oct 31. Aging Cell. 2020. PMID: 33128835 Free PMC article.
-
A new risk score based on twelve hepatocellular carcinoma-specific gene expression can predict the patients' prognosis.Aging (Albany NY). 2018 Sep 21;10(9):2480-2497. doi: 10.18632/aging.101563. Aging (Albany NY). 2018. PMID: 30243023 Free PMC article.
-
WAVE1 in neurons expressing the D1 dopamine receptor regulates cellular and behavioral actions of cocaine.Proc Natl Acad Sci U S A. 2017 Feb 7;114(6):1395-1400. doi: 10.1073/pnas.1621185114. Epub 2017 Jan 23. Proc Natl Acad Sci U S A. 2017. PMID: 28115704 Free PMC article.
-
Early blood immune molecular alterations in cynomolgus monkeys with a PSEN1 mutation causing familial Alzheimer's disease.Alzheimers Dement. 2024 Aug;20(8):5492-5510. doi: 10.1002/alz.14046. Epub 2024 Jul 7. Alzheimers Dement. 2024. PMID: 38973166 Free PMC article.
References
-
- Tanzi RE, Bertram L. Twenty years of the Alzheimer's disease amyloid hypothesis: a genetic perspective. Cell. 2005;120:545–555. - PubMed
-
- Ballard C, et al. Alzheimer's disease. Lancet. 2011;377:1019–1031. - PubMed
-
- Takenawa T, Suetsugu S. The WASP-WAVE protein network: connecting the membrane to the cytoskeleton. Nat. Rev. Mol. Cell Biol. 2007;8:37–48. - PubMed
-
- Eden S, Rohatgi R, Podtelejnikov AV, Mann M, Kirschner MW. Mechanism of regulation of WAVE1-induced actin nucleation by Rac1 and Nck. Nature. 2002;418:790–793. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

