The Role of Autophagy in Kidney Inflammatory Injury via the NF-κB Route Induced by LPS
- PMID: 26283886
- PMCID: PMC4532974
- DOI: 10.7150/ijms.12460
The Role of Autophagy in Kidney Inflammatory Injury via the NF-κB Route Induced by LPS
Abstract
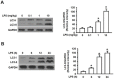
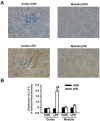
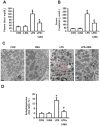
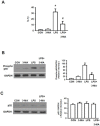
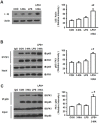
Acute kidney injury (AKI) is a systemic inflammatory response syndrome associated with poor clinical outcomes. No treatments effective for AKI are currently available. Thus, there is an urgent need of development of treatments effective for AKI. Autophagy, an intracellular proteolytic system, is induced in renal cells during AKI. However, whether autophagy is protective or injurious for AKI needs to be clearly clarified. We addressed this question by pharmacological inhibition of autophagy using a mouse model of lipopolysaccharide (LPS) induced-AKI. We found that autophagy was induced in renal cortex of mice during LPS-induced AKI as reflected by a dose-and time-dependent increased accumulation of light chain 3-II (LC3-II), the common marker of autophagy, compared to that of control group; 2) the occurrence of intensive, punctate and increased immunohistochemical staining image of LC3-II in renal cortex; 3) the significant increase in the expression levels of Beclin-1, another key marker of autophagy; 4) the significantly increased levels of plasma urea and serum creatinine and 5) the significant increase in autophagagosome area ratio. We observed that 3-methyladenine (3-MA), a pharmacological inhibitor of autophagy, blocked autophagy flux, alleviated AKI and protected against LPS-induced AKI. LPS triggered kidney inflammation by activation of the canonical NF-κB pathway. This route can be modulated by autophagy. Activation of the canonical NF-κB pathway was reduced in 3-MA+LPS as compared to that in LPS-treated group of mice. Mice pretreated with 3-MA before exposure to LPS showed a reduction in p65 phosphorylation, resulting in the accumulation of ubiquitinated IκB. In conclusion, impairment of autophagy ameliorates LPS-induced inflammation and decreases kidney injury. The accumulation of ubiquitinated IκB may be responsible for this effect.
Keywords: 3-methyladenine; IκB; LPS-induced kidney injury; autophagy; inflammation.
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures

References
-
- KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. Kidney Int. 2012;(Suppl 2):19–36. - PubMed
-
- Maddens B, Vandendriessche B, Demon D. et al. Severity of sepsis-induced acute kidney injury in a novel mouse model is age dependent. Crit Care Med. 2012;40:2638–46. - PubMed
-
- Nguyen HB, Rivers EP, Abrahamian FM. et al. Severe sepsis and septic shock: review of the literature and emergency department management guidelines. Ann Emerg Med. 2006;48:28–54. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

