β-Adrenergic receptor signaling and modulation of long-term potentiation in the mammalian hippocampus
- PMID: 26286656
- PMCID: PMC4561407
- DOI: 10.1101/lm.031088.113
β-Adrenergic receptor signaling and modulation of long-term potentiation in the mammalian hippocampus
Abstract
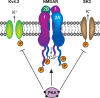
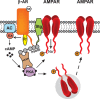
Encoding new information in the brain requires changes in synaptic strength. Neuromodulatory transmitters can facilitate synaptic plasticity by modifying the actions and expression of specific signaling cascades, transmitter receptors and their associated signaling complexes, genes, and effector proteins. One critical neuromodulator in the mammalian brain is norepinephrine (NE), which regulates multiple brain functions such as attention, perception, arousal, sleep, learning, and memory. The mammalian hippocampus receives noradrenergic innervation and hippocampal neurons express β-adrenergic receptors, which are known to play important roles in gating the induction of long-lasting forms of synaptic potentiation. These forms of long-term potentiation (LTP) are believed to importantly contribute to long-term storage of spatial and contextual memories in the brain. In this review, we highlight the contributions of noradrenergic signaling in general and β-adrenergic receptors in particular, toward modulating hippocampal LTP. We focus on the roles of NE and β-adrenergic receptors in altering the efficacies of specific signaling molecules such as NMDA and AMPA receptors, protein phosphatases, and translation initiation factors. Also, the roles of β-adrenergic receptors in regulating synaptic "tagging" and "capture" of LTP within synaptic networks of the hippocampus are reviewed. Understanding the molecular and cellular bases of noradrenergic signaling will enrich our grasp of how the brain makes new, enduring memories, and may shed light on credible strategies for improving mental health through treatment of specific disorders linked to perturbed memory processing and dysfunctional noradrenergic synaptic transmission.
© 2015 O'Dell et al.; Published by Cold Spring Harbor Laboratory Press.
Figures


References
-
- Abel T, Nguyen PV, Barad M, Deuel TA, Kandel ER, Bourtchouladze R. 1997. Genetic demonstration of a role for PKA in the late phase of LTP and in hippocampus-based long-term memory. Cell 88: 615–626. - PubMed
-
- Abraham WC. 2008. Metaplasticity: tuning synapses and networks for plasticity. Nat Rev Neurosci 9: 387. - PubMed
-
- Abraham WC, Bear MF. 1996. Metaplasticity: the plasticity of synaptic plasticity. Trends Neurosci 19: 126–130. - PubMed
-
- Abraham WC, Williams JM. 2008. LTP maintenance and its protein synthesis-dependence. Neurobiol Learn Mem 89: 260–268. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
