Effect of Brewing Duration on the Antioxidant and Hepatoprotective Abilities of Tea Phenolic and Alkaloid Compounds in a t-BHP Oxidative Stress-Induced Rat Hepatocyte Model
- PMID: 26287152
- PMCID: PMC6331988
- DOI: 10.3390/molecules200814985
Effect of Brewing Duration on the Antioxidant and Hepatoprotective Abilities of Tea Phenolic and Alkaloid Compounds in a t-BHP Oxidative Stress-Induced Rat Hepatocyte Model
Abstract
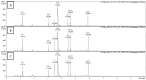
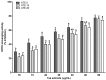
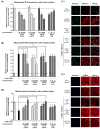
Tea is an interesting source of antioxidants capable of counteracting the oxidative stress implicated in liver diseases. We investigated the impact of antioxidant molecules provided by a mixture of teas' leaves (green, oolong, pu-erh) after different infusion durations in the prevention of oxidative stress in isolated rat hepatocytes, by comparison with pure epigallocatechin-3-gallate (EGCG), the main representative of tea catechins. Dried aqueous tea extracts (ATE) obtained after 5, 15 and 30 min infusion time were characterized for total polyphenols (gallic acid equivalent), catechins, gallic acid and caffeine (HPLC-DAD/ESI-MS) contents, and for scavenging ability against 2,2-diphenyl-1-picrylhydrazyl free radical. Hepatoprotection was evaluated through hepatocyte viability tests using tert-butyl hydroperoxide as a stress inducer, (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, neutral red uptake, real-time cellular impedance) and mitochondrial function tests. We showed that a 5-min incubation time is sufficient for an optimal bioaccessibility of tea compounds with the highest antioxidative ability, which decreases for longer durations. A 4-h pretreatment of cells with ATE significantly prevented cell death by regulating reactive oxygen species production and maintaining mitochondrial integrity. Pure EGCG, at doses similar in ATE (5-12 µM), was inefficient, suggesting a plausible synergy of several water-soluble tea compounds to explain the ATE beneficial effects.
Keywords: Camellia sinensis; EGCG; ROS; antioxidant; bioaccessibility; hepatocytes; mitochondrial membrane integrity; polyphenols; tea.
Conflict of interest statement
L. Braud’s Ph.D. student salary was funded by “Thés de la Pagode Company”. L. Peyre, G. de Sousa, M. Armand, R. Rahmani, and J-M. Maixent, declare no conflict of interest. The funding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Figures

Similar articles
-
Factors affecting the levels of tea polyphenols and caffeine in tea leaves.J Agric Food Chem. 2003 Mar 26;51(7):1864-73. doi: 10.1021/jf021066b. J Agric Food Chem. 2003. PMID: 12643643
-
Cytoprotection by almond skin extracts or catechins of hepatocyte cytotoxicity induced by hydroperoxide (oxidative stress model) versus glyoxal or methylglyoxal (carbonylation model).Chem Biol Interact. 2010 Apr 29;185(2):101-9. doi: 10.1016/j.cbi.2010.03.003. Epub 2010 Mar 6. Chem Biol Interact. 2010. PMID: 20211157
-
DHA sensitizes FaO cells to tert-BHP-induced oxidative effects. Protective role of EGCG.Food Chem Toxicol. 2013 Dec;62:750-7. doi: 10.1016/j.fct.2013.10.013. Epub 2013 Oct 16. Food Chem Toxicol. 2013. PMID: 24140970
-
The antioxidant and pro-oxidant activities of green tea polyphenols: a role in cancer prevention.Arch Biochem Biophys. 2010 Sep 1;501(1):65-72. doi: 10.1016/j.abb.2010.06.013. Epub 2010 Jun 15. Arch Biochem Biophys. 2010. PMID: 20558130 Free PMC article. Review.
-
Effects of green tea polyphenol extract and epigallocatechin-3-O-gallate on diabetes mellitus and diabetic complications: Recent advances.Crit Rev Food Sci Nutr. 2024;64(17):5719-5747. doi: 10.1080/10408398.2022.2157372. Epub 2022 Dec 19. Crit Rev Food Sci Nutr. 2024. PMID: 36533409 Review.
Cited by
-
Tea Polyphenols Relieve the Fluoride-Induced Oxidative Stress in the Intestinal Porcine Epithelial Cell Model.Toxics. 2025 Jan 24;13(2):83. doi: 10.3390/toxics13020083. Toxics. 2025. PMID: 39997898 Free PMC article.
-
Effects of Green Tea and Green Tea Incorporated in Nanoparticle Lyotropic Liquid Crystal on Exercise Adaptations: A High-Intensity Interval Training Pre-Clinical Study.Nutrients. 2022 Aug 7;14(15):3226. doi: 10.3390/nu14153226. Nutrients. 2022. PMID: 35956402 Free PMC article.
-
Possible Mechanisms of Dark Tea in Cancer Prevention and Management: A Comprehensive Review.Nutrients. 2023 Sep 7;15(18):3903. doi: 10.3390/nu15183903. Nutrients. 2023. PMID: 37764687 Free PMC article. Review.
-
Effects of Epigallocatechin Gallate on Tert-Butyl Hydroperoxide-Induced Mitochondrial Dysfunction in Rat Liver Mitochondria and Hepatocytes.Oxid Med Cell Longev. 2016;2016:7573131. doi: 10.1155/2016/7573131. Epub 2016 Dec 15. Oxid Med Cell Longev. 2016. PMID: 28074116 Free PMC article.
References
-
- Roberts E.A.H. The phenolic substances of manufactured tea. II.—Their origin as enzymic oxidation products in fermentation. J. Sci. Food Agric. 1958;9:212–216. doi: 10.1002/jsfa.2740090405. - DOI
-
- Peterson J., Dwyer J., Bhagwat S., Haytowitz D., Holden J., Eldridge A.L., Beecher G., Aladesanmi J. Major flavonoids in dry tea. J. Food Compos. Anal. 2005;18:487–501. doi: 10.1016/j.jfca.2004.05.006. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

