Iron Deficiency Impairs Intra-Hepatic Lymphocyte Mediated Immune Response
- PMID: 26287688
- PMCID: PMC4542211
- DOI: 10.1371/journal.pone.0136106
Iron Deficiency Impairs Intra-Hepatic Lymphocyte Mediated Immune Response
Abstract
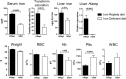
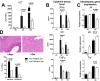
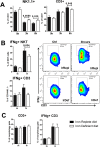
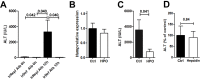
Hepatic expression of iron homeostasis genes and serum iron parameters predict the success of immunosuppression withdrawal following clinical liver transplantation, a phenomenon known as spontaneous operational tolerance. In experimental animal models, spontaneous liver allograft tolerance is established through a process that requires intra-hepatic lymphocyte activation and deletion. Our aim was to determine if changes in systemic iron status regulate intra-hepatic lymphocyte responses. We used a murine model of lymphocyte-mediated acute liver inflammation induced by Concanavalin A (ConA) injection employing mice fed with an iron-deficient (IrDef) or an iron-balanced diet (IrRepl). While the mild iron deficiency induced by the IrDef diet did not significantly modify the steady state immune cell repertoire and systemic cytokine levels, it significantly dampened inflammatory liver damage after ConA challenge. These findings were associated with a marked decrease in T cell and NKT cell activation following ConA injection in IrDef mice. The decreased liver injury observed in IrDef mice was independent from changes in the gut microflora, and was replicated employing an iron specific chelator that did not modify intra-hepatic hepcidin secretion. Furthermore, low-dose iron chelation markedly impaired the activation of isolated T cells in vitro. All together, these results suggest that small changes in iron homeostasis can have a major effect in the regulation of intra-hepatic lymphocyte mediated responses.
Conflict of interest statement
Figures


References
-
- Nairz M, Fritsche G, Crouch ML, Barton HC, Fang FC, Weiss G. Slc11a1 limits intracellular growth of Salmonella enterica sv. Typhimurium by promoting macrophage immune effector functions and impairing bacterial iron acquisition. Cellular microbiology. 2009;11(9):1365–81. 10.1111/j.1462-5822.2009.01337.x - DOI - PMC - PubMed
-
- Oexle H, Kaser A, Most J, Bellmann-Weiler R, Werner ER, Werner-Felmayer G, et al. Pathways for the regulation of interferon-gamma-inducible genes by iron in human monocytic cells. Journal of leukocyte biology. 2003;74(2):287–94. . - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

