Targeting the Checkpoint to Kill Cancer Cells
- PMID: 26295265
- PMCID: PMC4598780
- DOI: 10.3390/biom5031912
Targeting the Checkpoint to Kill Cancer Cells
Abstract
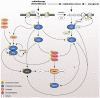
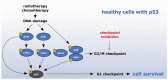
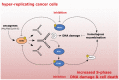
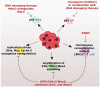
Cancer treatments such as radiotherapy and most of the chemotherapies act by damaging DNA of cancer cells. Upon DNA damage, cells stop proliferation at cell cycle checkpoints, which provides them time for DNA repair. Inhibiting the checkpoint allows entry to mitosis despite the presence of DNA damage and can lead to cell death. Importantly, as cancer cells exhibit increased levels of endogenous DNA damage due to an excessive replication stress, inhibiting the checkpoint kinases alone could act as a directed anti-cancer therapy. Here, we review the current status of inhibitors targeted towards the checkpoint effectors and discuss mechanisms of their actions in killing of cancer cells.
Keywords: ATM; ATR; Chk1; DNA damage response; Wee1; cancer; checkpoint; inhibitor; p53; replication stress.
Figures

Similar articles
-
ATR/CHK1 inhibitors and cancer therapy.Radiother Oncol. 2018 Mar;126(3):450-464. doi: 10.1016/j.radonc.2017.09.043. Epub 2017 Oct 18. Radiother Oncol. 2018. PMID: 29054375 Free PMC article. Review.
-
Targeting nuclear kinases in cancer: development of cell cycle kinase inhibitors.Pharmacol Ther. 2014 May;142(2):258-69. doi: 10.1016/j.pharmthera.2013.12.010. Epub 2013 Dec 19. Pharmacol Ther. 2014. PMID: 24362082 Review.
-
Targeting the S and G2 checkpoint to treat cancer.Drug Discov Today. 2012 Mar;17(5-6):194-202. doi: 10.1016/j.drudis.2011.12.009. Epub 2011 Dec 15. Drug Discov Today. 2012. PMID: 22192883 Review.
-
Molecular Pathways: Targeting ATR in Cancer Therapy.Clin Cancer Res. 2015 Nov 1;21(21):4780-5. doi: 10.1158/1078-0432.CCR-15-0479. Epub 2015 Sep 11. Clin Cancer Res. 2015. PMID: 26362996 Free PMC article. Review.
-
Targeting WEE1 Kinase in Cancer.Trends Pharmacol Sci. 2016 Oct;37(10):872-881. doi: 10.1016/j.tips.2016.06.006. Epub 2016 Jul 14. Trends Pharmacol Sci. 2016. PMID: 27427153 Review.
Cited by
-
Noninvasive PET Imaging of CDK4/6 Activation in Breast Cancer.J Nucl Med. 2020 Mar;61(3):437-442. doi: 10.2967/jnumed.119.232603. Epub 2019 Sep 3. J Nucl Med. 2020. PMID: 31481582 Free PMC article.
-
Alpinetin: A Review of Its Pharmacology and Pharmacokinetics.Front Pharmacol. 2022 Feb 4;13:814370. doi: 10.3389/fphar.2022.814370. eCollection 2022. Front Pharmacol. 2022. PMID: 35185569 Free PMC article. Review.
-
Alternative Chk1-independent S/M checkpoint in somatic cells that prevents premature mitotic entry.Med Oncol. 2017 Apr;34(4):70. doi: 10.1007/s12032-017-0932-3. Epub 2017 Mar 27. Med Oncol. 2017. PMID: 28349497
-
Acceleration or Brakes: Which Is Rational for Cell Cycle-Targeting Neuroblastoma Therapy?Biomolecules. 2021 May 18;11(5):750. doi: 10.3390/biom11050750. Biomolecules. 2021. PMID: 34069817 Free PMC article. Review.
-
Identification of Key Pathways and Genes in the Dynamic Progression of HCC Based on WGCNA.Genes (Basel). 2018 Feb 14;9(2):92. doi: 10.3390/genes9020092. Genes (Basel). 2018. PMID: 29443924 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

