Amyloid Precursor Protein (APP) Metabolites APP Intracellular Fragment (AICD), Aβ42, and Tau in Nuclear Roles
- PMID: 26296890
- PMCID: PMC4583011
- DOI: 10.1074/jbc.R115.677211
Amyloid Precursor Protein (APP) Metabolites APP Intracellular Fragment (AICD), Aβ42, and Tau in Nuclear Roles
Abstract
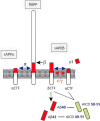
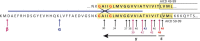
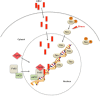
Amyloid precursor protein (APP) metabolites (amyloid-β (Aβ) peptides) and Tau are the main components of senile plaques and neurofibrillary tangles, the two histopathological hallmarks of Alzheimer disease. Consequently, intense research has focused upon deciphering their physiological roles to understand their altered state in Alzheimer disease pathophysiology. Recently, the impact of APP metabolites (APP intracellular fragment (AICD) and Aβ) and Tau on the nucleus has emerged as an important, new topic. Here we discuss (i) how AICD, Aβ, and Tau reach the nucleus and how AICD and Aβ control protein expression at the transcriptional level, (ii) post-translational modifications of AICD, Aβ, and Tau, and (iii) what these three molecules have in common.
Keywords: Alzheimer disease; DNA-protein interaction; Tau protein (Tau); amyloid precursor protein (APP); amyloid-beta (AB); gene regulation; gene transcription.
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures
References
-
- De Strooper B., Annaert W. (2010) Novel research horizons for presenilins and γ-secretases in cell biology and disease. Annu. Rev. Cell Dev. Biol. 26, 235–260 - PubMed
-
- Cleary J. P., Walsh D. M., Hofmeister J. J., Shankar G. M., Kuskowski M. A., Selkoe D. J., Ashe K. H. (2005) Natural oligomers of the amyloid-β protein specifically disrupt cognitive function. Nature Neurosci. 8, 79–84 - PubMed
-
- Kayed R., Glabe C. G. (2006) Conformation-dependent anti-amyloid oligomer antibodies. Methods Enzymol. 413, 326–344 - PubMed
-
- Lambert M. P., Barlow A. K., Chromy B. A., Edwards C., Freed R., Liosatos M., Morgan T. E., Rozovsky I., Trommer B., Viola K. L., Wals P., Zhang C., Finch C. E., Krafft G. A., Klein W. L. (1998) Diffusible, nonfibrillar ligands derived from Aβ1–42 are potent central nervous system neurotoxins. Proc. Natl. Acad. Sci. U.S.A. 95, 6448–6453 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

