Insulin-Producing Endocrine Cells Differentiated In Vitro From Human Embryonic Stem Cells Function in Macroencapsulation Devices In Vivo
- PMID: 26304037
- PMCID: PMC4572906
- DOI: 10.5966/sctm.2015-0079
Insulin-Producing Endocrine Cells Differentiated In Vitro From Human Embryonic Stem Cells Function in Macroencapsulation Devices In Vivo
Abstract
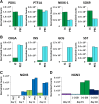
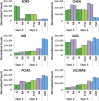
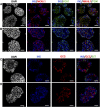
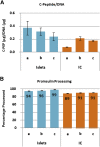
The PEC-01 cell population, differentiated from human embryonic stem cells (hESCs), contains pancreatic progenitors (PPs) that, when loaded into macroencapsulation devices (to produce the VC-01 candidate product) and transplanted into mice, can mature into glucose-responsive insulin-secreting cells and other pancreatic endocrine cells involved in glucose metabolism. We modified the protocol for making PEC-01 cells such that 73%-80% of the cell population consisted of PDX1-positive (PDX1+) and NKX6.1+ PPs. The PPs were further differentiated to islet-like cells (ICs) that reproducibly contained 73%-89% endocrine cells, of which approximately 40%-50% expressed insulin. A large fraction of these insulin-positive cells were single hormone-positive and expressed the transcription factors PDX1 and NKX6.1. To preclude a significant contribution of progenitors to the in vivo function of ICs, we used a simple enrichment process to remove remaining PPs, yielding aggregates that contained 93%-98% endocrine cells and 1%-3% progenitors. Enriched ICs, when encapsulated and implanted into mice, functioned similarly to the VC-01 candidate product, demonstrating conclusively that in vitro-produced hESC-derived insulin-producing cells can mature and function in vivo in devices. A scaled version of our suspension culture was used, and the endocrine aggregates could be cryopreserved and retain functionality. Although ICs expressed multiple important β cell genes, the cells contained relatively low levels of several maturity-associated markers. Correlating with this, the time to function of ICs was similar to PEC-01 cells, indicating that ICs required cell-autonomous maturation after delivery in vivo, which would occur concurrently with graft integration into the host.
Significance: Type 1 diabetes (T1D) affects approximately 1.25 million people in the U.S. alone and is deadly if not managed with insulin injections. This paper describes the production of insulin-producing cells in vitro and a new protocol for producing the cells, representing another potential cell source for a diabetes cell therapy. These cells can be loaded into a protective device that is implanted under the skin. The device is designed to protect the cells from immune rejection by the implant recipient. The implant can engraft and respond to glucose by secreting insulin, thus potentially replacing the β cells lost in patients with T1D.
Keywords: Cell therapy; Diabetes; Drug delivery systems; Embryonic stem cells; Insulin-secreting cells; Pancreas development.
©AlphaMed Press.
Figures


Similar articles
-
Enrichment of human embryonic stem cell-derived NKX6.1-expressing pancreatic progenitor cells accelerates the maturation of insulin-secreting cells in vivo.Stem Cells. 2013 Nov;31(11):2432-42. doi: 10.1002/stem.1489. Stem Cells. 2013. PMID: 23897760
-
Establishment of a rapid and footprint-free protocol for differentiation of human embryonic stem cells into pancreatic endocrine cells with synthetic mRNAs encoding transcription factors.Stem Cell Res Ther. 2018 Oct 25;9(1):277. doi: 10.1186/s13287-018-1038-3. Stem Cell Res Ther. 2018. PMID: 30359326 Free PMC article.
-
Generation of insulin-producing islet-like clusters from human embryonic stem cells.Stem Cells. 2007 Aug;25(8):1940-53. doi: 10.1634/stemcells.2006-0761. Epub 2007 May 17. Stem Cells. 2007. PMID: 17510217
-
In vitro generation of pancreatic β-cells for diabetes treatment. I. β-like cells derived from human pluripotent stem cells.Folia Histochem Cytobiol. 2019;57(1):1-14. doi: 10.5603/FHC.a2019.0001. Epub 2019 Mar 14. Folia Histochem Cytobiol. 2019. PMID: 30869153 Review.
-
Regenerative medicine for diabetes: differentiation of human pluripotent stem cells into functional β-cells in vitro and their proposed journey to clinical translation.Vitam Horm. 2014;95:223-48. doi: 10.1016/B978-0-12-800174-5.00009-0. Vitam Horm. 2014. PMID: 24559920 Review.
Cited by
-
Strategies to Improve the Safety of iPSC-Derived β Cells for β Cell Replacement in Diabetes.Transpl Int. 2022 Aug 24;35:10575. doi: 10.3389/ti.2022.10575. eCollection 2022. Transpl Int. 2022. PMID: 36090777 Free PMC article. Review.
-
V-Maf Musculoaponeurotic Fibrosarcoma Oncogene Homolog A Synthetic Modified mRNA Drives Reprogramming of Human Pancreatic Duct-Derived Cells Into Insulin-Secreting Cells.Stem Cells Transl Med. 2016 Nov;5(11):1525-1537. doi: 10.5966/sctm.2015-0318. Epub 2016 Jul 12. Stem Cells Transl Med. 2016. PMID: 27405779 Free PMC article.
-
Generation of Multipotent Stem Cells from Adult Human Peripheral Blood Following the Treatment with Platelet-Derived Mitochondria.Cells. 2020 May 29;9(6):1350. doi: 10.3390/cells9061350. Cells. 2020. PMID: 32485922 Free PMC article.
-
Three-dimensional bioprinting of functional β-islet-like constructs.Int J Bioprint. 2023 Jan 9;9(2):665. doi: 10.18063/ijb.v9i2.665. eCollection 2023. Int J Bioprint. 2023. PMID: 37065656 Free PMC article.
-
Status of islet transplantation and innovations to sustainable outcomes: novel sites, cell sources, and drug delivery strategies.Front Transplant. 2024 Nov 1;3:1485444. doi: 10.3389/frtra.2024.1485444. eCollection 2024. Front Transplant. 2024. PMID: 39553396 Free PMC article. Review.
References
-
- Kelly OG, Chan MY, Martinson LA, et al. Cell-surface markers for the isolation of pancreatic cell types derived from human embryonic stem cells. Nat Biotechnol. 2011;29:750–756. - PubMed
-
- Kroon E, Martinson LA, Kadoya K, et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat Biotechnol. 2008;26:443–452. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

