Electronic Trigger-Based Intervention to Reduce Delays in Diagnostic Evaluation for Cancer: A Cluster Randomized Controlled Trial
- PMID: 26304875
- PMCID: PMC4622097
- DOI: 10.1200/JCO.2015.61.1301
Electronic Trigger-Based Intervention to Reduce Delays in Diagnostic Evaluation for Cancer: A Cluster Randomized Controlled Trial
Abstract
Purpose: We tested whether prospective use of electronic health record-based trigger algorithms to identify patients at risk of diagnostic delays could prevent delays in diagnostic evaluation for cancer.
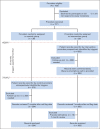
Methods: We performed a cluster randomized controlled trial of primary care providers (PCPs) at two sites to test whether triggers that prospectively identify patients with potential delays in diagnostic evaluation for lung, colorectal, or prostate cancer can reduce time to follow-up diagnostic evaluation. Intervention steps included queries of the electronic health record repository for patients with abnormal findings and lack of associated follow-up actions, manual review of triggered records, and communication of this information to PCPs via secure e-mail and, if needed, phone calls to ensure message receipt. We compared times to diagnostic evaluation and proportions of patients followed up between intervention and control cohorts based on final review at 7 months.
Results: We recruited 72 PCPs (36 in the intervention group and 36 in the control group) and applied the trigger to all patients under their care from April 20, 2011, to July 19, 2012. Of 10,673 patients with abnormal findings, the trigger flagged 1,256 patients (11.8%) as high risk for delayed diagnostic evaluation. Times to diagnostic evaluation were significantly lower in intervention patients compared with control patients flagged by the colorectal trigger (median, 104 v 200 days, respectively; n = 557; P < .001) and prostate trigger (40% received evaluation at 144 v 192 days, respectively; n = 157; P < .001) but not the lung trigger (median, 65 v 93 days, respectively; n = 19; P = .59). More intervention patients than control patients received diagnostic evaluation by final review (73.4% v 52.2%, respectively; relative risk, 1.41; 95% CI, 1.25 to 1.58).
Conclusion: Electronic trigger-based interventions seem to be effective in reducing time to diagnostic evaluation of colorectal and prostate cancer as well as improving the proportion of patients who receive follow-up. Similar interventions could improve timeliness of diagnosis of other serious conditions.
Trial registration: ClinicalTrials.gov NCT01346839.
© 2015 by American Society of Clinical Oncology.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest are found in the article online at
Figures

References
-
- Markides GA, Newman CM. Medical malpractice claims in relation to colorectal malignancy in the National Health Service. Colorectal Dis. 2014;16:48–56. - PubMed
-
- Gandhi TK, Kachalia A, Thomas EJ, et al. Missed and delayed diagnoses in the ambulatory setting: A study of closed malpractice claims. Ann Intern Med. 2006;145:488–496. - PubMed
-
- Schiff GD, Puopolo AL, Huben-Kearney A, et al. Primary care closed claims experience of Massachusetts malpractice insurers. JAMA Intern Med. 2013;173:2063–2068. - PubMed
-
- Savage JR, Weiner GM. Litigation in otolaryngology: Trends and recommendations. J Laryngol Otol. 2006;120:1001–1004. - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

