Infectious hematopoietic necrosis virus (IHNV) persistence in Sockeye Salmon: influence on brain transcriptome and subsequent response to the viral mimic poly(I:C)
- PMID: 26306576
- PMCID: PMC4549833
- DOI: 10.1186/s12864-015-1759-y
Infectious hematopoietic necrosis virus (IHNV) persistence in Sockeye Salmon: influence on brain transcriptome and subsequent response to the viral mimic poly(I:C)
Abstract
Background: Sockeye Salmon are an iconic species widely distributed throughout the North Pacific. A devastating pathogen of Sockeye Salmon is infectious hematopoietic necrosis virus (IHNV, genus Novirhabdovirus, family Rhabdoviridae). It has been postulated that IHNV is maintained in salmon populations by persisting over the life of its host and/or by residing in natural reservoirs other than its susceptible hosts. Herein we demonstrate the presence of IHNV in the brain of Sockeye Salmon that survived an experimentally-induced outbreak, suggesting the presence of viral persistence in this susceptible species. To understand the viral persistent state in Sockeye Salmon we profiled the transcriptome to evaluate the host response in asymptomatic carriers and to determine what effects (if any) IHNV exposure may have on subsequent virus challenges.
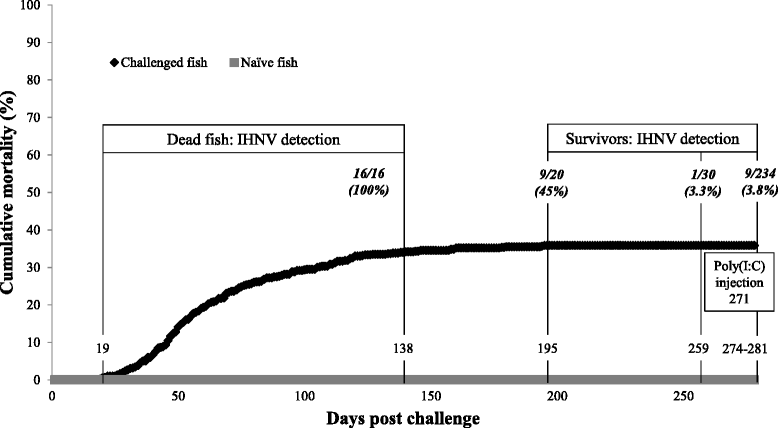
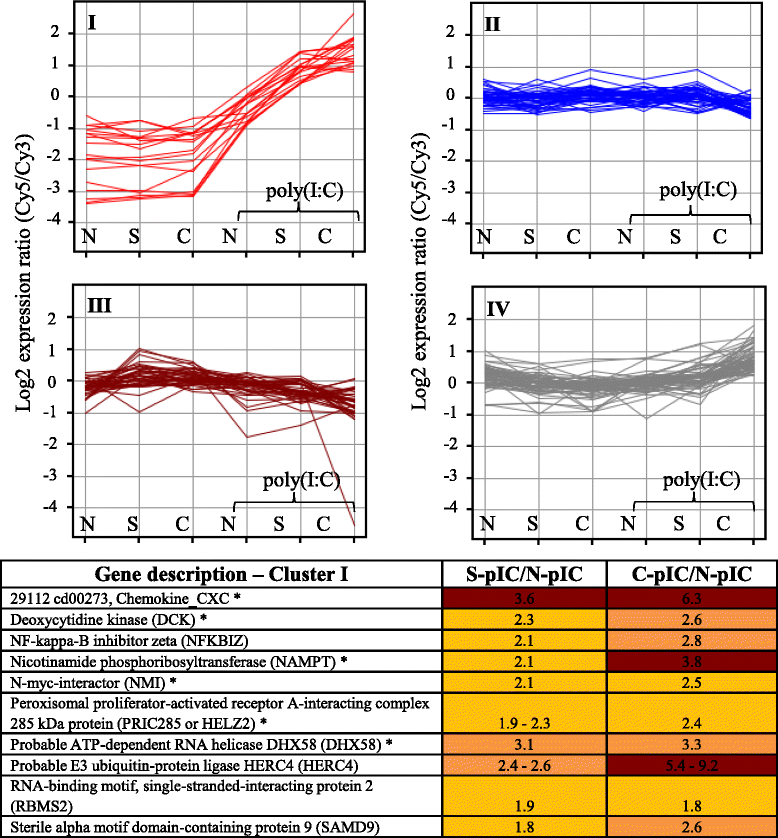
Results: A laboratory disease model to simulate a natural IHNV outbreak in Sockeye Salmon resulted in over a third of the population incurring acute IHN disease and mortality during the first four months after initial exposure. Nine months post IHNV exposure, despite the absence of disease and mortality, a small percentage (<4 %) of the surviving population contained IHNV in brain. Transcriptome analysis in brain of asymptomatic virus carriers and survivors without virus exhibited distinct transcriptional profiles in comparison to naïve fish. Characteristic for carriers was the up-regulation of genes involved in antibody production and antigen presentation. In both carriers and survivors a down-regulation of genes related to cholesterol biosynthesis, resembling an antiviral mechanism observed in higher vertebrates was revealed along with differences in nervous system development. Moreover, following challenge with poly(I:C), survivors and carriers displayed an elevated antiviral immune response in comparison to naïve fish.
Conclusions: IHN virus persistence was identified in Sockeye Salmon where it elicited a unique brain transcriptome profile suggesting an ongoing adaptive immune response. IHNV carriers remained uncompromised in mounting efficient innate antiviral responses when exposed to a viral mimic. The capacity of IHNV to reside in asymptomatic hosts supports a virus carrier hypothesis and if proven infectious, could have significant epidemiological consequences towards maintaining and spreading IHNV among susceptible host populations.
Figures

Similar articles
-
Sockeye salmon demonstrate robust yet distinct transcriptomic kidney responses to rhabdovirus (IHNV) exposure and infection.Fish Shellfish Immunol. 2019 Nov;94:525-538. doi: 10.1016/j.fsi.2019.09.042. Epub 2019 Sep 17. Fish Shellfish Immunol. 2019. PMID: 31539572
-
De novo assembly of Sockeye salmon kidney transcriptomes reveal a limited early response to piscine reovirus with or without infectious hematopoietic necrosis virus superinfection.BMC Genomics. 2016 Nov 2;17(1):848. doi: 10.1186/s12864-016-3196-y. BMC Genomics. 2016. PMID: 27806699 Free PMC article.
-
Transmission potential of infectious hematopoietic necrosis virus in APEX-IHN®-vaccinated Atlantic salmon.Dis Aquat Organ. 2017 Jan 24;122(3):213-221. doi: 10.3354/dao03076. Dis Aquat Organ. 2017. PMID: 28117300
-
The antiviral innate immune response in fish: evolution and conservation of the IFN system.J Mol Biol. 2013 Dec 13;425(24):4904-20. doi: 10.1016/j.jmb.2013.09.033. Epub 2013 Sep 27. J Mol Biol. 2013. PMID: 24075867 Review.
-
Licensed DNA Vaccines against Infectious Hematopoietic Necrosis Virus (IHNV).Recent Pat DNA Gene Seq. 2013 Apr 1;7(1):62-5. doi: 10.2174/1872215611307010009. Recent Pat DNA Gene Seq. 2013. PMID: 22670604 Review.
Cited by
-
Antiviral Actions of 25-Hydroxycholesterol in Fish Vary With the Virus-Host Combination.Front Immunol. 2021 Feb 24;12:581786. doi: 10.3389/fimmu.2021.581786. eCollection 2021. Front Immunol. 2021. PMID: 33717065 Free PMC article.
-
Nervous Necrosis Virus Modulation of European Sea Bass (Dicentrarchus labrax, L.) Immune Genes and Transcriptome towards Establishment of Virus Carrier State.Int J Mol Sci. 2023 Nov 22;24(23):16613. doi: 10.3390/ijms242316613. Int J Mol Sci. 2023. PMID: 38068937 Free PMC article.
-
Fish Brain Cell Lines Can Be Infected with Adenoviral Vectors and Support Transgene Expression-An In Vitro Approach.Int J Mol Sci. 2024 Dec 12;25(24):13357. doi: 10.3390/ijms252413357. Int J Mol Sci. 2024. PMID: 39769121 Free PMC article.
-
Transcriptomic Profiling of the Adaptive and Innate Immune Responses of Atlantic Salmon to Renibacterium salmoninarum Infection.Front Immunol. 2020 Oct 28;11:567838. doi: 10.3389/fimmu.2020.567838. eCollection 2020. Front Immunol. 2020. PMID: 33193341 Free PMC article.
-
Synergistic osmoregulatory dysfunction during salmon lice (Lepeophtheirus salmonis) and infectious hematopoietic necrosis virus co-infection in sockeye salmon (Oncorhynchus nerka) smolts.J Fish Dis. 2019 Jun;42(6):869-882. doi: 10.1111/jfd.12989. Epub 2019 Apr 12. J Fish Dis. 2019. PMID: 30977528 Free PMC article.
References
-
- Burgner RL. Life History of Sockeye Salmon (Oncorhynchus nerka) In: Groot CLM, editor. Pacific Salmon Life Histories. Vancouver: UBC Press; 1991.
-
- Meyers TR, Korn D, Burton TM, Glass K, Follett JE, Thomas JB, et al. Infectious hematopoietic necrosis virus (IHNV) in Alaskan sockeye salmon culture from 1973 to 2000: Annual virus prevalences and titers in broodstocks compared with juvenile losses. J Aquat Anim Health. 2003;15(1):21–30. doi: 10.1577/1548-8667(2003)015<0021:IHNVII>2.0.CO;2. - DOI
-
- Dietzgen RG, Calisher CH, Kurath G, Kuzmin IV, Rodriguez LL, Stone DM, et al. Family Rhabdoviridae. In: King AMQ, Adams MJ, Carstens EB, Lefkowitz EJ, et al., editors. Virus Taxonomy: Classification and Nomenclature of Viruses: Ninth Report of the International Committee on Taxonomy of Viruses. London: Elsevier Academic Press; 2012.
-
- Rucker RR, Whipple WJ, Parvin JR, Evans CA. A contagious disease of sockeye salmon possibly of virus origin. US Fish Wildlife Service Fisheries Bulletin. 1953;54:35–46.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

