The Contingency of Cocaine Administration Accounts for Structural and Functional Medial Prefrontal Deficits and Increased Adrenocortical Activation
- PMID: 26311772
- PMCID: PMC4549401
- DOI: 10.1523/JNEUROSCI.4961-14.2015
The Contingency of Cocaine Administration Accounts for Structural and Functional Medial Prefrontal Deficits and Increased Adrenocortical Activation
Erratum in
- J Neurosci. 2015 Dec 2;35(48):16012
Abstract
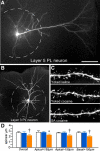
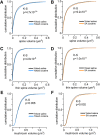
The prelimbic region (PL) of the medial prefrontal cortex (mPFC) is implicated in the relapse of drug-seeking behavior. Optimal mPFC functioning relies on synaptic connections involving dendritic spines in pyramidal neurons, whereas prefrontal dysfunction resulting from elevated glucocorticoids, stress, aging, and mental illness are each linked to decreased apical dendritic branching and spine density in pyramidal neurons in these cortical fields. The fact that cocaine use induces activation of the stress-responsive hypothalamo-pituitary-adrenal axis raises the possibility that cocaine-related impairments in mPFC functioning may be manifested by similar changes in neuronal architecture in mPFC. Nevertheless, previous studies have generally identified increases, rather than decreases, in structural plasticity in mPFC after cocaine self-administration. Here, we use 3D imaging and analysis of dendritic spine morphometry to show that chronic cocaine self-administration leads to mild decreases of apical dendritic branching, prominent dendritic spine attrition in PL pyramidal neurons, and working memory deficits. Importantly, these impairments were largely accounted for in groups of rats that self-administered cocaine compared with yoked-cocaine- and saline-matched counterparts. Follow-up experiments failed to demonstrate any effects of either experimenter-administered cocaine or food self-administration on structural alterations in PL neurons. Finally, we verified that the cocaine self-administration group was distinguished by more protracted increases in adrenocortical activity compared with yoked-cocaine- and saline-matched controls. These studies suggest a mechanism whereby increased adrenocortical activity resulting from chronic cocaine self-administration may contribute to regressive prefrontal structural and functional plasticity.
Significance statement: Stress, aging, and mental illness are each linked to decreased prefrontal plasticity. Here, we show that chronic cocaine self-administration in rats leads to decrements in medial prefrontal structural and functional plasticity. Notably, these impairments were largely accounted for in rats that self-administered cocaine compared with yoked counterparts. Moreover, we verified previous reports showing that adrenocortical output is augmented by cocaine administration and is more protracted in rats that were permitted to receive the drug contingently instead of passively. These studies suggest that increased adrenocortical activity resulting from cocaine self-administration may contribute to regressive prefrontal structural and functional plasticity.
Keywords: HPA; corticosterone; dendritic spine; prelimbic; self-administration; working memory.
Copyright © 2015 the authors 0270-6474/15/3511897-14$15.00/0.
Figures









References
-
- Broadbear JH, Winger G, Cicero TJ, Woods JH. Effects of response contingent and noncontingent cocaine injection on hypothalamic-pituitary-adrenal activity in rhesus monkeys. J Pharmacol Exp Ther. 1999;290:393–402. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
