Myelin Proteolipid Protein Complexes with αv Integrin and AMPA Receptors In Vivo and Regulates AMPA-Dependent Oligodendrocyte Progenitor Cell Migration through the Modulation of Cell-Surface GluR2 Expression
- PMID: 26311781
- PMCID: PMC4549408
- DOI: 10.1523/JNEUROSCI.5151-14.2015
Myelin Proteolipid Protein Complexes with αv Integrin and AMPA Receptors In Vivo and Regulates AMPA-Dependent Oligodendrocyte Progenitor Cell Migration through the Modulation of Cell-Surface GluR2 Expression
Abstract
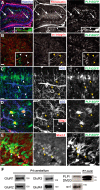
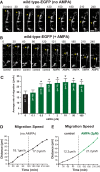
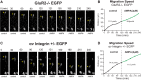
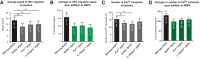
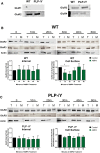
In previous studies, stimulation of ionotropic AMPA/kainate glutamate receptors on cultured oligodendrocyte cells induced the formation of a signaling complex that includes the AMPA receptor, integrins, calcium-binding proteins, and, surprisingly, the myelin proteolipid protein (PLP). AMPA stimulation of cultured oligodendrocyte progenitor cells (OPCs) also caused an increase in OPC migration. The current studies focused primarily on the formation of the PLP-αv integrin-AMPA receptor complex in vivo and whether complex formation impacts OPC migration in the brain. We found that in wild-type cerebellum, PLP associates with αv integrin and the calcium-impermeable GluR2 subunit of the AMPA receptor, but in mice lacking PLP, αv integrin did not associate with GluR2. Live imaging studies of OPC migration in ex vivo cerebellar slices demonstrated altered OPC migratory responses to neurotransmitter stimulation in the absence of PLP and GluR2 or when αv integrin levels were reduced. Chemotaxis assays of purified OPCs revealed that AMPA stimulation was neither attractive nor repulsive but clearly increased the migration rate of wild-type but not PLP null OPCs. AMPA receptor stimulation of wild-type OPCs caused decreased cell-surface expression of the GluR2 AMPA receptor subunit and increased intracellular Ca(2+) signaling, whereas PLP null OPCs did not reduce GluR2 at the cell surface or increase Ca(2+) signaling in response to AMPA treatment. Together, these studies demonstrate that PLP is critical for OPC responses to glutamate signaling and has important implications for OPC responses when levels of glutamate are high in the extracellular space, such as following demyelination.
Significance statement: After demyelination, such as occurs in multiple sclerosis, remyelination of axons is often incomplete, leading to loss of neuronal function and clinical disability. Remyelination may fail because oligodendrocyte precursor cells (OPCs) do not completely migrate into demyelinated areas or OPCs in lesions may not mature into myelinating oligodendrocytes. We have found that the myelin proteolipid protein is critical to regulating OPC migratory responses to the neurotransmitter glutamate through modulation of cell-surface expression of the calcium-impermeable GluR2 subunit of the AMPA glutamate receptor and increased intercellular Ca(2+) signaling. Altered glutamate homeostasis has been reported in demyelinated lesions. Therefore, understanding how OPCs respond to glutamate has important implications for treatment after white matter injury and disease.
Keywords: AMPA; GluR2; integrin; migration; myelin proteolipid protein; oligodendrocyte.
Copyright © 2015 the authors 0270-6474/15/3512018-15$15.00/0.
Figures




References
-
- Barres BA, Lazar MA, Raff MC. A novel role for thyroid hormone, glucocorticoids and retinoic acid in timing oligodendrocyte development. Development. 1994;120:1097–1108. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
