International Union of Basic and Clinical Pharmacology. XCIX. Angiotensin Receptors: Interpreters of Pathophysiological Angiotensinergic Stimuli [corrected]
- PMID: 26315714
- PMCID: PMC4630565
- DOI: 10.1124/pr.114.010454
International Union of Basic and Clinical Pharmacology. XCIX. Angiotensin Receptors: Interpreters of Pathophysiological Angiotensinergic Stimuli [corrected]
Erratum in
-
Correction to "Angiotensin Receptors: Interpreters of Pathophysiological Angiotensinergic Stimuli".Pharmacol Rev. 2015 Oct;67(4):820. doi: 10.1124/pr.114.010454err. Pharmacol Rev. 2015. PMID: 26335847 Free PMC article. No abstract available.
Abstract
The renin angiotensin system (RAS) produced hormone peptides regulate many vital body functions. Dysfunctional signaling by receptors for RAS peptides leads to pathologic states. Nearly half of humanity today would likely benefit from modern drugs targeting these receptors. The receptors for RAS peptides consist of three G-protein-coupled receptors—the angiotensin II type 1 receptor (AT1 receptor), the angiotensin II type 2 receptor (AT2 receptor), the MAS receptor—and a type II trans-membrane zinc protein—the candidate angiotensin IV receptor (AngIV binding site). The prorenin receptor is a relatively new contender for consideration, but is not included here because the role of prorenin receptor as an independent endocrine mediator is presently unclear. The full spectrum of biologic characteristics of these receptors is still evolving, but there is evidence establishing unique roles of each receptor in cardiovascular, hemodynamic, neurologic, renal, and endothelial functions, as well as in cell proliferation, survival, matrix-cell interaction, and inflammation. Therapeutic agents targeted to these receptors are either in active use in clinical intervention of major common diseases or under evaluation for repurposing in many other disorders. Broad-spectrum influence these receptors produce in complex pathophysiological context in our body highlights their role as precise interpreters of distinctive angiotensinergic peptide cues. This review article summarizes findings published in the last 15 years on the structure, pharmacology, signaling, physiology, and disease states related to angiotensin receptors. We also discuss the challenges the pharmacologist presently faces in formally accepting newer members as established angiotensin receptors and emphasize necessary future developments.
Copyright © 2015 by The American Society for Pharmacology and Experimental Therapeutics.
Figures
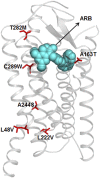
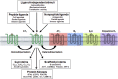
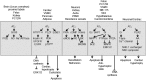

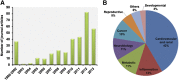
References
-
- Abadir PM, Periasamy A, Carey RM, Siragy HM. (2006) Angiotensin II type 2 receptor-bradykinin B2 receptor functional heterodimerization. Hypertension 48:316–322. - PubMed
-
- Abbasi S, Su B, Kellems RE, Yang J, Xia Y. (2005) The essential role of MEKK3 signaling in angiotensin II-induced calcineurin/nuclear factor of activated T-cells activation. J Biol Chem 280:36737–36746. - PubMed
-
- AbdAlla S, Abdel-Baset A, Lother H, el Massiery A, Quitterer U. (2005) Mesangial AT1/B2 receptor heterodimers contribute to angiotensin II hyperresponsiveness in experimental hypertension. J Mol Neurosci 26:185–192. - PubMed
-
- AbdAlla S, Lother H, Abdel-tawab AM, Quitterer U. (2001a) The angiotensin II AT2 receptor is an AT1 receptor antagonist. J Biol Chem 276:39721–39726. - PubMed
-
- AbdAlla S, Lother H, el Massiery A, Quitterer U. (2001b) Increased AT(1) receptor heterodimers in preeclampsia mediate enhanced angiotensin II responsiveness. Nat Med 7:1003–1009. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

