In vivo assessment of two endothelialization approaches on bioprosthetic valves for the treatment of chronic deep venous insufficiency
- PMID: 26316151
- PMCID: PMC6592699
- DOI: 10.1002/jbm.b.33507
In vivo assessment of two endothelialization approaches on bioprosthetic valves for the treatment of chronic deep venous insufficiency
Abstract
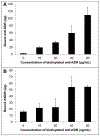
Chronic deep venous insufficiency is a debilitating disease with limited therapeutic interventions. A bioprosthetic venous valve could not only replace a diseased valve, but has the potential to fully integrate into the patient with a minimally invasive procedure. Previous work with valves constructed from small intestinal submucosa (SIS) showed improvements in patients' symptoms in clinical studies; however, substantial thickening of the implanted valve leaflets also occurred. As endothelial cells are key regulators of vascular homeostasis, their presence on the SIS valves may reduce the observed thickening. This work tested an off-the-shelf approach to capture circulating endothelial cells in vivo using biotinylated antikinase insert domain receptor antibodies in a suspended leaflet ovine model. The antibodies on SIS were oriented to promote cell capture and showed positive binding to endothelial cells in vitro; however, no differences were observed in leaflet thickness in vivo between antibody-modified and unmodified SIS. In an alternative approach, valves were pre-seeded with autologous endothelial cells and tested in vivo. Nearly all the implanted pre-seeded valves were patent and functioning; however, no statistical difference was observed in valve thickness with cell pre-seeding. Additional cell capture schemes or surface modifications should be examined to find an optimal method for encouraging SIS valve endothelialization to improve long-term valve function in vivo. © 2015 Wiley Periodicals, Inc. J Biomed Mater Res Part B: Appl Biomater, 104B: 1610-1621, 2016.
Keywords: endothelial cells; small intestinal submucosa (SIS); vascular endothelial growth factor receptor-2; venous insufficiency; venous valve.
© 2015 Wiley Periodicals, Inc.
Figures

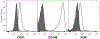

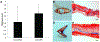

References
-
- Kaplan RM, Criqui MH, Denenberg JO, Bergan J, Fronek A. Quality of life in patients with chronic venous disease: San Diego population study. J Vasc Surg. 2003;37:1047–53. - PubMed
-
- Eberhardt RT, Raffetto JD. Chronic venous insufficiency. Circulation. 2005;111:2398–409. - PubMed
-
- Gloviczki P, Comerota AJ, Dalsing MC, Eklof BG, Gillespie DL, Gloviczki ML, Lohr JM, McLafferty RB, Meissner MH, Murad MH, Padberg FT, Pappas PJ, Passman MA, Raffetto JD, Vasquez MA, Wakefield TW. The care of patients with varicose veins and associated chronic venous diseases: Clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg. 2011;53:2S–48S. - PubMed
-
- Geerts W, Bergqvist D. Prevention of Venous Thromboembolism American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. CHEST. 2008;133:381S–453S. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

