Porous titanium bases for osteochondral tissue engineering
- PMID: 26320541
- PMCID: PMC4698168
- DOI: 10.1016/j.actbio.2015.08.045
Porous titanium bases for osteochondral tissue engineering
Abstract
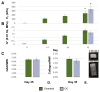
Tissue engineering of osteochondral grafts may offer a cell-based alternative to native allografts, which are in short supply. Previous studies promote the fabrication of grafts consisting of a viable cell-seeded hydrogel integrated atop a porous, bone-like metal. Advantages of the manufacturing process have led to the evaluation of porous titanium as the bone-like base material. Here, porous titanium was shown to support the growth of cartilage to produce native levels of Young's modulus, using a clinically relevant cell source. Mechanical and biochemical properties were similar or higher for the osteochondral constructs compared to chondral-only controls. Further investigation into the mechanical influence of the base on the composite material suggests that underlying pores may decrease interstitial fluid pressurization and applied strains, which may be overcome by alterations to the base structure. Future studies aim to optimize titanium-based tissue engineered osteochondral constructs to best match the structural architecture and strength of native grafts.
Statement of significance: The studies described in this manuscript follow up on previous studies from our lab pertaining to the fabrication of osteochondral grafts that consist of a bone-like porous metal and a chondrocyte-seeded hydrogel. Here, tissue engineered osteochondral grafts were cultured to native stiffness using adult chondrocytes, a clinically relevant cell source, and a porous titanium base, a material currently used in clinical implants. This porous titanium is manufactured via selective laser melting, offering the advantages of precise control over shape, pore size, and orientation. Additionally, this manuscript describes the mechanical influence of the porous base, which may have applicability to porous bases derived from other materials.
Keywords: Articular cartilage; Osteochondral grafts; Porous titanium; Tissue engineering.
Copyright © 2015. Published by Elsevier Ltd.
Figures



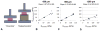




References
-
- Buckwalter JA, Mankin HJ. Articular cartilage repair and transplantation. Arthritis Rheum. 1998;41:1331–1342. - PubMed
-
- Cole BJ, Pascual-Garrido C, Grumet RC. Surgical management of articular cartilage defects in the knee. J Bone Joint Surg Am. 2009;91:1778–1790. - PubMed
-
- Flanigan DC, Harris JD, Trinh TQ, Siston RA, Brophy RH. Prevalence of chondral defects in athletes’ knees: a systematic review. Med Sci Sports Exerc. 2010;42:1795–1801. - PubMed
-
- Sherman SL, Garrity J, Bauer K, Cook J, Stannard J, Bugbee W. Fresh osteochondral allograft transplantation for the knee: current concepts. J Am Acad Orthop Surg. 2014;22:121–133. - PubMed
-
- Gortz S, Bugbee WD. Allografts in articular cartilage repair. J Bone Joint Surg Am. 2006;88:1374–1384. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

