Sustained Impairments in Brain Insulin/IGF Signaling in Adolescent Rats Subjected to Binge Alcohol Exposures during Development
- PMID: 26322248
- PMCID: PMC4550301
- DOI: 10.4172/2161-0681.1000106
Sustained Impairments in Brain Insulin/IGF Signaling in Adolescent Rats Subjected to Binge Alcohol Exposures during Development
Abstract
Background: Chronic or binge ethanol exposures during development can cause fetal alcohol spectrum disorder (FASD) which consists of an array of neurobehavioral deficits, together with structural, molecular, biochemical, and neurotransmitter abnormalities in the brain. Previous studies showed that perinatal neurodevelopmental defects in FASD are associated with inhibition of brain insulin and insulin-like growth factor (IGF) signaling. However, it is not known whether sustained abnormalities in adolescent brain structure and function are mediated by the same phenomena.
Aims: Using an early postnatal (3rd trimester equivalent) binge ethanol exposure model, we assessed neurobehavioral function, structure, and the integrity of insulin/IGF signaling in young adolescent cerebella.
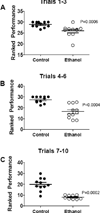
Methods: Long Evans male rats were treated with 50 µl of saline (vehicle) or 2 mg/kg of ethanol by i.p. injection on postnatal days (P) 2, 4, 6, and 8. On P19-20, rats were subjected to rotarod testing of motor function, and on P30, they were sacrificed to harvest cerebella for histological, molecular, and biochemical studies.
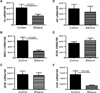
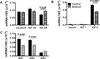
Results: Binge ethanol exposures impaired motor function, caused sustained cerebellar hypocellularity, and reduced neuronal and oligodendrocyte gene expression. These effects were associated with significant deficits in insulin and IGF signaling, including impaired receptor binding, reduced Akt, and increased GSK-3β activation.
Conclusions: FASD-associated neurobehavioral, structural, and functional abnormalities in young adolescent brains may be mediated by sustained inhibition of insulin/IGF-1 signaling needed for cell survival, neuronal plasticity, and myelin maintenance.
Keywords: Adolescence; Brain development; Brain insulin resistance; Central nervous system; Cerebellum; Fetal alcohol syndrome; Insulin signaling; Motor function; Receptor binding.
Figures




References
-
- Riley EP, McGee CL. Fetal alcohol spectrum disorders: an overview with emphasis on changes in brain and behavior. Exp Biol Med (Maywood) 2005;230:357–365. - PubMed
-
- Aronson M, Hagberg B. Neuropsychological disorders in children exposed to alcohol during pregnancy: a follow-up study of 24 children to alcoholic mothers in Göteborg, Sweden. Alcohol Clin Exp Res. 1998;22:321–324. - PubMed
-
- Kaplan-Estrin M, Jacobson SW, Jacobson JL. Neurobehavioral effects of prenatal alcohol exposure at 26 months. Neurotoxicol Teratol. 1999;21:503–511. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
