Compound A Inhibits Bladder Cancer Growth Predominantly via Glucocorticoid Receptor Transrepression
- PMID: 26322830
- PMCID: PMC5414678
- DOI: 10.1210/me.2015-1128
Compound A Inhibits Bladder Cancer Growth Predominantly via Glucocorticoid Receptor Transrepression
Abstract
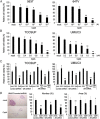
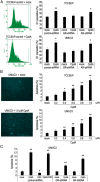
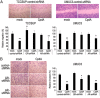
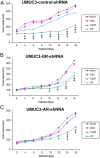
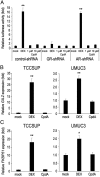
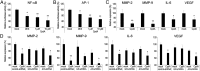
Recent evidence indicates that glucocorticoids (GCs) suppress bladder cancer cell invasion through the GC receptor (GR) pathway, whereas androgen-mediated androgen receptor (AR) signals induce bladder tumor progression. In this study, we assessed the effects of 2-(4-acetoxyphenyl)-2-chloro-N-methyl-ethylammonium chloride (compound A [CpdA]), which was shown to function as not only a GR modulator but also an AR antagonist, on the growth of bladder cancer. In GR/AR-positive cells, CpdA strongly inhibited cell proliferation and colony formation as well as increased G1 phase-arrested cell population and apoptosis. Specifically, CpdA at 1μM decreased cell viability of TCCSUP/UMUC3-control-short hairpin RNA (shRNA), TCCSUP/UMUC3-GR-shRNA, and TCCSUP/UMUC3-AR-shRNA by 50%/67%, 25%/26%, and 38%/58%, respectively. CpdA also inhibited cell migration and invasion of GR/AR-positive (up to 61% decrease) and GR-positive/AR-silencing (up to 51% decrease) lines and, less strongly, those of GR-silencing/AR-positive lines (up to 35% decrease). Additionally, in UMUC3-control xenograft-bearing male mice, CpdA more strongly suppressed tumor growth than dexamethasone or hydroxyflutamide. In reporter gene assays, CpdA failed to induce GR transactivation, whereas it antagonized dihydrotestosterone-enhanced AR transactivation. In contrast, CpdA reduced nuclear factor (NF)-κB and activator protein 1 transcriptional activities, indicating induction of GR-mediated transrepression. Correspondingly, the expression of NF-κB-related molecules, matrix metalloproteinase-2, matrix metalloproteinase-9, interleukin-6, and vascular endothelial growth factor, was significantly down-regulated by CpdA in control lines but not in GR-silencing cells. Moreover, coimmunoprecipitation showed that CpdA promoted the interactions between GR and NF-κB. Thus, CpdA likely inhibits bladder cancer growth predominantly via inducing GR transrepression and at least partially mediated through the AR pathway, suggesting its effects more beneficial than GCs/pure GR ligands or AR antagonists.
Figures

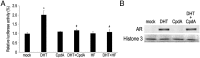
References
-
- Miyamoto H, Zheng Y, Izumi K. Nuclear hormone receptor signals as new therapeutic targets for urothelial carcinoma. Curr Cancer Drug Tar. 2012;12:14–22. - PubMed
-
- Zheng Y, Izumi K, Li Y, Ishiguro H, Miyamoto H. Contrary regulation of bladder cancer cell proliferation and invasion by dexamethasone-mediated glucocorticoid receptor signals. Mol Cancer Ther. 2012;11:2621–2632. - PubMed
-
- Zhang C, Wenger T, Mattern J, et al. Clinical and mechanistic aspects of glucocorticoid-induced chemotherapy resistance in the majority of solid tumors. Cancer Biol Ther. 2007;6:278–287. - PubMed
-
- Ishiguro H, Kawahara T, Zheng Y, Kashiwagi E, Li Y, Miyamoto H. Differential regulation of bladder cancer growth by various glucocorticoids: corticosterone and prednisone inhibit cell invasion without promoting cell proliferation or reducing cisplatin cytotoxicity. Cancer Chemother Pharmacol. 2014;74:249–255. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

