The contribution of Chlamydia-specific CD8⁺ T cells to upper genital tract pathology
- PMID: 26323581
- PMCID: PMC4747851
- DOI: 10.1038/icb.2015.74
The contribution of Chlamydia-specific CD8⁺ T cells to upper genital tract pathology
Abstract
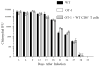
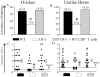
Genital chlamydial infections lead to severe upper reproductive tract pathology in a subset of untreated women. We demonstrated previously that tumor necrosis factor (TNF)-α-producing CD8(+) T cells contribute significantly to chlamydial upper genital tract pathology in female mice. In addition, we observed that minimal chlamydial oviduct pathology develops in OT-1 transgenic (OT-1) mice, wherein the CD8(+) T-cell repertoire is restricted to recognition of the ovalbumin peptide Ova(257-264), suggesting that non-Chlamydia-specific CD8(+) T cells may not be responsible for chlamydial pathogenesis. In the current study, we evaluated whether antigen-specific CD8(+) T cells mediate chlamydial pathology. Groups of wild-type (WT) C57BL/6J, OT-1 mice, and OT-1 mice replete with WT CD8(+) T cells (1 × 10(6) cells per mouse intravenously) were infected intravaginally with C. muridarum (5 × 10(4) IFU/mouse). Serum total anti-Chlamydia antibody and total splenic anti-Chlamydia interferon (IFN)-γ and TNF-α responses were comparable among the three groups of animals. However, Chlamydia-specific IFN-γ and TNF-α production from purified splenic CD8(+) T cells of OT-1 mice was minimal, whereas responses in OT-1 mice replete with WT CD8(+) T cells were comparable to those in WT animals. Vaginal chlamydial clearance was comparable between the three groups of mice. Importantly, the incidence and severity of oviduct and uterine horn pathology was significantly reduced in OT-1 mice but reverted to WT levels in OT-1 mice replete with WT CD8(+) T cells. Collectively, these results demonstrate that Chlamydia-specific CD8(+) T cells contribute significantly to upper genital tract pathology.
Conflict of interest statement
Conflict of interest: The authors declare no conflict of interest.
Figures

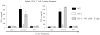
References
-
- Belland R, Ojcius DM, Byrne GI. Chlamydia Nat Rev Microbiol. 2004;2:530–531. - PubMed
-
- Westrom L, Joesoef R, Reynolds G, Hagdu A, Thompson SE. Pelvic inflammatory disease and fertility. A cohort study of 1,844 women with laparoscopically verified disease and 657 control women with normal laparoscopic results. Sex Transm Dis. 1992;19:185–192. - PubMed
-
- Westrom L, Mardh PA. Chlamydial salpingitis. Br Med Bull. 1983;39:145–150. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

