The caveolin-cavin system plays a conserved and critical role in mechanoprotection of skeletal muscle
- PMID: 26323694
- PMCID: PMC4555827
- DOI: 10.1083/jcb.201501046
The caveolin-cavin system plays a conserved and critical role in mechanoprotection of skeletal muscle
Abstract
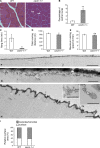
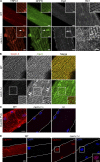
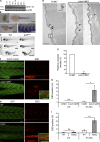
Dysfunction of caveolae is involved in human muscle disease, although the underlying molecular mechanisms remain unclear. In this paper, we have functionally characterized mouse and zebrafish models of caveolae-associated muscle disease. Using electron tomography, we quantitatively defined the unique three-dimensional membrane architecture of the mature muscle surface. Caveolae occupied around 50% of the sarcolemmal area predominantly assembled into multilobed rosettes. These rosettes were preferentially disassembled in response to increased membrane tension. Caveola-deficient cavin-1(-/-) muscle fibers showed a striking loss of sarcolemmal organization, aberrant T-tubule structures, and increased sensitivity to membrane tension, which was rescued by muscle-specific Cavin-1 reexpression. In vivo imaging of live zebrafish embryos revealed that loss of muscle-specific Cavin-1 or expression of a dystrophy-associated Caveolin-3 mutant both led to sarcolemmal damage but only in response to vigorous muscle activity. Our findings define a conserved and critical role in mechanoprotection for the unique membrane architecture generated by the caveolin-cavin system.
© 2015 Lo et al.
Figures




Comment in
-
Mechanoprotection by skeletal muscle caveolae.Bioarchitecture. 2016;6(1):22-7. doi: 10.1080/19490992.2015.1131891. Bioarchitecture. 2016. PMID: 26760312 Free PMC article.
References
-
- Berghmans S., Murphey R.D., Wienholds E., Neuberg D., Kutok J.L., Fletcher C.D., Morris J.P., Liu T.X., Schulte-Merker S., Kanki J.P., et al. 2005. tp53 mutant zebrafish develop malignant peripheral nerve sheath tumors. Proc. Natl. Acad. Sci. USA. 102:407–412. 10.1073/pnas.0406252102 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

