Predicting the integration of overlapping memories by decoding mnemonic processing states during learning
- PMID: 26327243
- PMCID: PMC4651766
- DOI: 10.1016/j.neuroimage.2015.08.051
Predicting the integration of overlapping memories by decoding mnemonic processing states during learning
Abstract
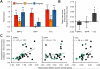
The hippocampal memory system is thought to alternate between two opposing processing states: encoding and retrieval. When present experience overlaps with past experience, this creates a potential tradeoff between encoding the present and retrieving the past. This tradeoff may be resolved by memory integration-that is, by forming a mnemonic representation that links present experience with overlapping past experience. Here, we used fMRI decoding analyses to predict when - and establish how - past and present experiences become integrated in memory. In an initial experiment, we alternately instructed subjects to adopt encoding, retrieval or integration states during overlapping learning. We then trained across-subject pattern classifiers to 'read out' the instructed processing states from fMRI activity patterns. We show that an integration state was clearly dissociable from encoding or retrieval states. Moreover, trial-by-trial fluctuations in decoded evidence for an integration state during learning reliably predicted behavioral expressions of successful memory integration. Strikingly, the decoding algorithm also successfully predicted specific instances of spontaneous memory integration in an entirely independent sample of subjects for whom processing state instructions were not administered. Finally, we show that medial prefrontal cortex and hippocampus differentially contribute to encoding, retrieval, and integration states: whereas hippocampus signals the tradeoff between encoding vs. retrieval states, medial prefrontal cortex actively represents past experience in relation to new learning.
Keywords: Episodic memory; Hippocampus; Integration; MVPA; Medial prefrontal cortex; Reinstatement.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures




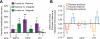
References
-
- Anderson MC, McCulloch KC. Integration as a general boundary condition on retrieval-induced forgetting. Journal of Experimental Psychology: Learning, Memory, and Cognition. 1999;25(3):608. Retrieved from Google Scholar.
-
- Bor D, Duncan J, Wiseman RJ, Owen AM. Encoding strategies dissociate prefrontal activity from working memory demand. Neuron. 2003;37(2):361–7. - PubMed
-
- Buzsáki G. Two-stage model of memory trace formation: A role for “noisy” brain states. Neuroscience. 1989;31(3):551–70. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

