Differential Protein Expression in Congenital and Acquired Cholesteatomas
- PMID: 26335306
- PMCID: PMC4559438
- DOI: 10.1371/journal.pone.0137011
Differential Protein Expression in Congenital and Acquired Cholesteatomas
Abstract
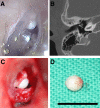
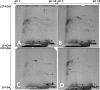
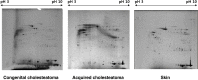
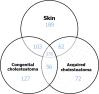
Congenital cholesteatomas are epithelial lesions that present as an epithelial pearl behind an intact eardrum. Congenital and acquired cholesteatomas progress quite differently from each other and progress patterns can provide clues about the unique origin and pathogenesis of the abnormality. However, the exact pathogenic mechanisms by which cholesteatomas develop remain unknown. In this study, key proteins that directly affect cholesteatoma pathogenesis are investigated with proteomics and immunohistochemistry. Congenital cholesteatoma matrices and retroauricular skin were harvested during surgery in 4 patients diagnosed with a congenital cholesteatoma. Tissue was also harvested from the retraction pocket in an additional 2 patients during middle ear surgery. We performed 2-dimensional (2D) electrophoresis to detect and analyze spots that are expressed only in congenital cholesteatoma and matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF/MS) to separate proteins by molecular weight. Protein expression was confirmed by immunohistochemical staining. The image analysis of 2D electrophoresis showed that 4 congenital cholesteatoma samples had very similar protein expression patterns and that 127 spots were exclusively expressed in congenital cholesteatomas. Of these 127 spots, 10 major spots revealed the presence of titin, forkhead transcription activator homolog (FKH 5-3), plectin 1, keratin 10, and leucine zipper protein 5 by MALDI-TOF/MS analysis. Immunohistochemical staining showed that FKH 5-3 and titin were expressed in congenital cholesteatoma matrices, but not in acquired cholesteatomas. Our study shows that protein expression patterns are completely different in congenital cholesteatomas, acquired cholesteatomas, and skin. Moreover, non-epithelial proteins, including FKH 5-3 and titin, were unexpectedly expressed in congenital cholesteatoma tissue. Our data indicates that congenital cholesteatoma origins may differ from those of acquired cholesteatomas, which originate from retraction pocket epithelia.
Conflict of interest statement
Figures


Similar articles
-
Comparative analysis of the expression of E-cadherin, β-catenin, and β1 integrin in congenital and acquired cholesteatoma.Eur Arch Otorhinolaryngol. 2016 Apr;273(4):845-51. doi: 10.1007/s00405-015-3621-x. Epub 2015 Apr 12. Eur Arch Otorhinolaryngol. 2016. PMID: 25864182
-
[Congenital cholesteatomas in Japanese--forty from our experience and fifty-five from a survey of the Japanese literature].Nihon Jibiinkoka Gakkai Kaiho. 1996 Sep;99(9):1200-7. Nihon Jibiinkoka Gakkai Kaiho. 1996. PMID: 8914417 Review. Japanese.
-
Differences in dendritic cells in congenital and acquired cholesteatomas.Laryngoscope. 1993 Nov;103(11 Pt 1):1214-7. doi: 10.1288/00005537-199311000-00002. Laryngoscope. 1993. PMID: 8231573
-
Immunohistochemical analysis of the cytokeratin expression in middle ear cholesteatoma and related epithelial tissues.Ann Otol Rhinol Laryngol. 1992 Nov;101(11):931-8. doi: 10.1177/000348949210101109. Ann Otol Rhinol Laryngol. 1992. PMID: 1280020
-
Increased numbers of mast cells in human middle ear cholesteatomas: implications for treatment.Am J Otol. 1998 May;19(3):266-72. Am J Otol. 1998. PMID: 9596172 Review.
References
-
- Maniu A, Harabagiu O, Perde Schrepler M, Catana A, Fanuta B, Mogoanta CA. Molecular biology of cholesteatoma. Rom J Morphol Embryo. 2014;55(1):7–13. - PubMed
-
- Frankel S, Berson S, Godwin T, Han JC, Parisier SC. Differences in dendritic cells in congenital and acquired cholesteatomas. Laryngoscope. 1993;103(11 Pt 1):1214–1217 - PubMed
-
- Olszewska E, Rutkowska J, Minovi A, Sieskiewicz A, Rogowski M, Dazert S. The Role of p21 and p53 Proteins in Congenital Cholesteatoma. Otol Neurotology. 2013;34(2):266–274. - PubMed
-
- Kojima H, Miyazaki H, Shiwa M, Tanaka Y, Moriyama H. Molecular biological diagnosis of congenital and acquired cholesteatoma on the basis of differences in telomere length. Laryngoscope. 2001;111(5):867–873. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

