Molecular Regulation of Parturition: A Myometrial Perspective
- PMID: 26337112
- PMCID: PMC4632865
- DOI: 10.1101/cshperspect.a023069
Molecular Regulation of Parturition: A Myometrial Perspective
Abstract
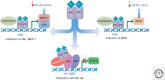
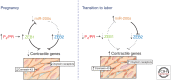
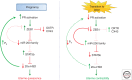
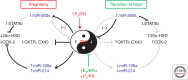
The molecular mechanisms that maintain quiescence of the myometrium throughout most of pregnancy and promote its transformation to a highly coordinated contractile unit culminating in labor are complex and intertwined. During pregnancy, progesterone (P4) produced by the placenta and/or ovary serves a dominant role in maintaining myometrial quiescence by blocking proinflammatory response pathways and expression of so-called "contractile" genes. In the majority of placental mammals, increased uterine contractility near term is heralded by an increase in circulating estradiol-17β (E2) and/or increased estrogen receptor α (ERα) activity and a sharp decline in circulating P4 levels. However, in women, circulating levels of P4 and progesterone receptors (PR) in myometrium remain elevated throughout pregnancy and into labor. This has led to the concept that increased uterine contractility leading to term and preterm labor is mediated, in part, by a decline in PR function. The biochemical mechanisms for this decrease in PR function are also multifaceted and interwoven. In this paper, we focus on the molecular mechanisms that mediate myometrial quiescence and contractility and their regulation by the two central hormones of pregnancy, P4 and estradiol-17β. The integrative roles of microRNAs also are considered.
Copyright © 2015 Cold Spring Harbor Laboratory Press; all rights reserved.
Figures
Similar articles
-
Multifactorial Regulation of Myometrial Contractility During Pregnancy and Parturition.Front Endocrinol (Lausanne). 2019 Oct 25;10:714. doi: 10.3389/fendo.2019.00714. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 31708868 Free PMC article. Review.
-
Myometrial progesterone responsiveness and the control of human parturition.J Soc Gynecol Investig. 2004 May;11(4):193-202. doi: 10.1016/j.jsgi.2003.12.004. J Soc Gynecol Investig. 2004. PMID: 15120691 Review.
-
MicroRNAs--mediators of myometrial contractility during pregnancy and labour.Nat Rev Endocrinol. 2013 Jul;9(7):391-401. doi: 10.1038/nrendo.2013.96. Epub 2013 May 14. Nat Rev Endocrinol. 2013. PMID: 23669656 Review.
-
Reciprocal Feedback Between miR-181a and E2/ERα in Myometrium Enhances Inflammation Leading to Labor.J Clin Endocrinol Metab. 2016 Oct;101(10):3646-3656. doi: 10.1210/jc.2016-2078. Epub 2016 Jul 26. J Clin Endocrinol Metab. 2016. PMID: 27459534 Free PMC article.
-
Steroid hormone control of myometrial contractility and parturition.Semin Cell Dev Biol. 2007 Jun;18(3):321-31. doi: 10.1016/j.semcdb.2007.05.003. Epub 2007 May 18. Semin Cell Dev Biol. 2007. PMID: 17613262 Review.
Cited by
-
Transcriptional control of parturition: insights from gene regulation studies in the myometrium.Mol Hum Reprod. 2021 May 8;27(5):gaab024. doi: 10.1093/molehr/gaab024. Mol Hum Reprod. 2021. PMID: 33823545 Free PMC article. Review.
-
Progesterone and its receptor signaling in cervical remodeling: Mechanisms of physiological actions and therapeutic implications.J Steroid Biochem Mol Biol. 2022 Oct;223:106137. doi: 10.1016/j.jsbmb.2022.106137. Epub 2022 Jun 8. J Steroid Biochem Mol Biol. 2022. PMID: 35690241 Free PMC article. Review.
-
Pro-inflammatory signals induce 20α-HSD expression in myometrial cells: A key mechanism for local progesterone withdrawal.J Cell Mol Med. 2021 Jul;25(14):6773-6785. doi: 10.1111/jcmm.16681. Epub 2021 Jun 10. J Cell Mol Med. 2021. PMID: 34114342 Free PMC article.
-
Multifactorial Regulation of Myometrial Contractility During Pregnancy and Parturition.Front Endocrinol (Lausanne). 2019 Oct 25;10:714. doi: 10.3389/fendo.2019.00714. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 31708868 Free PMC article. Review.
-
Characterization of an Adapted Murine Model of Intrauterine Inflammation-Induced Preterm Birth.Am J Pathol. 2020 Feb;190(2):295-305. doi: 10.1016/j.ajpath.2019.10.013. Epub 2019 Dec 16. Am J Pathol. 2020. PMID: 31837289 Free PMC article.
References
-
- Auphan N, DiDonato JA, Rosette C, Helmberg A, Karin M. 1995. Immunosuppression by glucocorticoids: Inhibition of NF-κB activity through induction of IκB synthesis. Science 270: 286–290. - PubMed
-
- Baldwin ASJ. 1996. The NF-κB and IκB proteins: New discoveries and insights. Annu Rev Immunol 14: 649–683. - PubMed
-
- Blencowe H, Cousens S, Oestergaard MZ, Chou D, Moller AB, Narwal R, Adler A, Vera GC, Rohde S, Say L, et al. 2012. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: A systematic analysis and implications. Lancet 379: 2162–2172. - PubMed
-
- Boldin MP, Baltimore D. 2012. MicroRNAs, new effectors and regulators of NF-κB. Immunol Rev 246: 205–220. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
