Infiltrating bone marrow mesenchymal stem cells (BM-MSCs) increase prostate cancer cell invasion via altering the CCL5/HIF2α/androgen receptor signals
- PMID: 26342197
- PMCID: PMC4695008
- DOI: 10.18632/oncotarget.4515
Infiltrating bone marrow mesenchymal stem cells (BM-MSCs) increase prostate cancer cell invasion via altering the CCL5/HIF2α/androgen receptor signals
Abstract
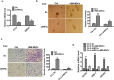
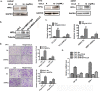
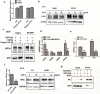
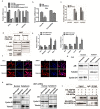
Several infiltrating cells in the tumor microenvironment could influence the cancer progression via secreting various cytokines. Here, we found the CCL5 secreted from BM-MSCs suppressed androgen receptor (AR) signals via enhancing the expression of hypoxia inducible factor 2α (HIF2α) in prostate cancer (PCa) cells. Mechanism dissection revealed that the increased HIF2α might alter the AR-HSP90 interaction to suppress the AR transactivation, and inhibition of HIF2α reversed the BM-MSCs-increased PCa stem cell population and PCa cells invasion. Importantly, CCL5 could suppress the prolyl hydroxylases (PHDs) expression, which might then lead to suppress VHL-mediated HIF2α ubiquitination. Together, these results demonstrated that the CCL5 signals from infiltrating BM-MSC cells to HIF2α signals within PCa cells might play a key role to increase PCa stem cell population and PCa metastasis via altering the AR signals. Targeting this newly identified CCL5/HIF2α/AR axis signal axis may allow us to develop a novel way to suppress PCa metastasis.
Keywords: CCL5; HIF2α; androgen receptor; bone marrow mesenchymal stem cells; prostate cancer.
Conflict of interest statement
The authors have no conflicts of interests to declare.
Figures

Similar articles
-
Infiltrating bone marrow mesenchymal stem cells increase prostate cancer stem cell population and metastatic ability via secreting cytokines to suppress androgen receptor signaling.Oncogene. 2014 May 22;33(21):2768-78. doi: 10.1038/onc.2013.233. Epub 2013 Jun 24. Oncogene. 2014. PMID: 23792449
-
Infiltrating mast cells enhance prostate cancer invasion via altering LncRNA-HOTAIR/PRC2-androgen receptor (AR)-MMP9 signals and increased stem/progenitor cell population.Oncotarget. 2015 Jun 10;6(16):14179-90. doi: 10.18632/oncotarget.3651. Oncotarget. 2015. PMID: 25895025 Free PMC article.
-
Infiltrated pre-adipocytes increase prostate cancer metastasis via modulation of the miR-301a/androgen receptor (AR)/TGF-β1/Smad/MMP9 signals.Oncotarget. 2015 May 20;6(14):12326-39. doi: 10.18632/oncotarget.3619. Oncotarget. 2015. PMID: 25940439 Free PMC article.
-
Concise review: androgen receptor differential roles in stem/progenitor cells including prostate, embryonic, stromal, and hematopoietic lineages.Stem Cells. 2014 Sep;32(9):2299-308. doi: 10.1002/stem.1722. Stem Cells. 2014. PMID: 24740898 Review.
-
Hypoxia and hypoxia inducible factors in cancer stem cell maintenance.Curr Top Microbiol Immunol. 2010;345:21-30. doi: 10.1007/82_2010_75. Curr Top Microbiol Immunol. 2010. PMID: 20582533 Review.
Cited by
-
Prostate cancer-specific proinflammatory cytokines and chemokines impact on cancer stem cell development, lineage plasticity and heterogeneity in an Ancestral/racially diverse population: review.Cancer Metastasis Rev. 2025 Mar 19;44(2):41. doi: 10.1007/s10555-025-10259-1. Cancer Metastasis Rev. 2025. PMID: 40106077 Review.
-
Suppressive Role of Androgen/Androgen Receptor Signaling via Chemokines on Prostate Cancer Cells.J Clin Med. 2019 Mar 13;8(3):354. doi: 10.3390/jcm8030354. J Clin Med. 2019. PMID: 30871130 Free PMC article. Review.
-
Osteoid cell-derived chemokines drive bone-metastatic prostate cancer.Front Oncol. 2023 Mar 21;13:1100585. doi: 10.3389/fonc.2023.1100585. eCollection 2023. Front Oncol. 2023. PMID: 37025604 Free PMC article. Review.
-
Mouse models of metastasis: progress and prospects.Dis Model Mech. 2017 Sep 1;10(9):1061-1074. doi: 10.1242/dmm.030403. Dis Model Mech. 2017. PMID: 28883015 Free PMC article. Review.
-
Self-assembling nanoparticles encapsulating zoledronic acid inhibit mesenchymal stromal cells differentiation, migration and secretion of proangiogenic factors and their interactions with prostate cancer cells.Oncotarget. 2017 Jun 27;8(26):42926-42938. doi: 10.18632/oncotarget.17216. Oncotarget. 2017. PMID: 28477013 Free PMC article.
References
-
- Zhu P, Baek S.H, Bourk E.M, Ohgi K.A, Garcia-Bassets I, Sanjo H, Akira S, Kotol P.F, Glass C.K, Rosenfeld M.G, et al. Macrophage/cancer cell interactions mediate hormone resistance by a nuclear receptor derepression pathway. Cell. 2006;124:615–629. - PubMed
-
- Zeng Y, Opeskin K, Goad J, Williams E.D. Tumor-induced activation of lymphatic endothelial cells via vascular endothelial growth factor receptor-2 is critical for prostate cancer lymphatic metastasis. Cancer Res. 2006;66:9566–9575. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

