Evaluation of the International Prognostic Score (IPS-7) and a Simpler Prognostic Score (IPS-3) for advanced Hodgkin lymphoma in the modern era
- PMID: 26343802
- PMCID: PMC4881845
- DOI: 10.1111/bjh.13634
Evaluation of the International Prognostic Score (IPS-7) and a Simpler Prognostic Score (IPS-3) for advanced Hodgkin lymphoma in the modern era
Abstract
The International Prognostic Score (IPS-7) is the most commonly used risk stratification tool for advanced Hodgkin lymphoma (HL), however recent studies suggest the IPS-7 is less discriminating due to improved outcomes with contemporary therapy. We evaluated the seven variables for IPS-7 recorded at study entry for 854 patients enrolled on Eastern Cooperative Oncology Group 2496 trial. Univariate and multivariate Cox models were used to assess their prognostic ability for freedom from progression (FFP) and overall survival (OS). The IPS-7 remained prognostic however its prognostic range has narrowed. On multivariate analysis, two factors (age, stage) remained significant for FFP and three factors (age, stage, haemoglobin level) for OS. An alternative prognostic index, the IPS-3, was constructed using age, stage and haemoglobin level, which provided four distinct risk groups [FFP (P = 0·0001) and OS (P < 0·0001)]. IPS-3 outperformed the IPS-7 on risk prediction for both FFP and OS by model fit and discrimination criteria. Using reclassification calibration, 18% of IPS-7 low risk patients were re-classified as intermediate risk and 13% of IPS-7 intermediate risk patients as low risk. For patients with advanced HL, the IPS-3 may provide a simpler and more accurate framework for risk assessment in the modern era. Validation of these findings in other large data sets is planned.
Keywords: ABVD; Hodgkin lymphoma; International Prognostic Score; Stanford V; prognostic score.
© 2015 John Wiley & Sons Ltd.
Conflict of interest statement
HL: no conflicts of interest to disclose
FH: no conflicts of interest to disclose
LG: no conflicts of interest to disclose
RF: no conflicts of interest to disclose
NB: no conflicts of interest to disclose
MC: no conflicts of interest to disclose
RD: no conflicts of interest to disclose
HW: no conflicts of interest to disclose
PS: no conflicts of interest to disclose
BC: no conflicts of interest to disclose
DS: no conflicts of interest to disclose
BK: no conflicts of interest to disclose
JF: no conflicts of interest to disclose
KB: no conflicts of interest to disclose
TH: no conflicts of interest to disclose
JT: no conflicts of interest to disclose
RH: no conflicts of interest to disclose
SH: no conflicts of interest to disclose
RA: no conflicts of interest to disclose
Figures

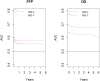
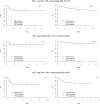
Comment in
-
Comments on the value International Prognostic Score (IPS), stage IV and age over 45 years, for the outcome of advanced Hodgkin lymphoma in chemotherapy containing adriamycin. The experience of the Spanish Hodgkin Lymphoma Study Group.Br J Haematol. 2017 Jul;178(2):336-338. doi: 10.1111/bjh.14127. Epub 2016 May 2. Br J Haematol. 2017. PMID: 27135208 No abstract available.
Similar articles
-
[Feasibility study of application of international prognostic score on prediction of prognosis for advanced Hodgkin's lymphoma].Ai Zheng. 2006 Aug;25(8):1013-8. Ai Zheng. 2006. PMID: 16965685 Chinese.
-
Serum levels of soluble CD30 improve International Prognostic Score in predicting the outcome of advanced Hodgkin's lymphoma.Ann Oncol. 2002 Dec;13(12):1908-14. doi: 10.1093/annonc/mdf333. Ann Oncol. 2002. PMID: 12453859
-
ABVD versus modified stanford V versus MOPPEBVCAD with optional and limited radiotherapy in intermediate- and advanced-stage Hodgkin's lymphoma: final results of a multicenter randomized trial by the Intergruppo Italiano Linfomi.J Clin Oncol. 2005 Dec 20;23(36):9198-207. doi: 10.1200/JCO.2005.02.907. Epub 2005 Sep 19. J Clin Oncol. 2005. PMID: 16172458 Clinical Trial.
-
What is the treatment of choice for advanced-stage Hodgkin's lymphoma: ABVD, Stanford V, or BEACOPP?Curr Hematol Rep. 2004 Jan;3(1):17-26. Curr Hematol Rep. 2004. PMID: 14695845 Review.
-
Standard therapy of advanced Hodgkin lymphoma.Hematology Am Soc Hematol Educ Program. 2009:497-506. doi: 10.1182/asheducation-2009.1.497. Hematology Am Soc Hematol Educ Program. 2009. PMID: 20008235 Review.
Cited by
-
The International Prognostic Score and HIV status predict red cell concentrate transfusion needs in Hodgkin lymphoma.Leuk Lymphoma. 2023 Mar;64(3):613-620. doi: 10.1080/10428194.2022.2157214. Epub 2022 Dec 23. Leuk Lymphoma. 2023. PMID: 36562564 Free PMC article.
-
The validation of advanced-stage Hodgkin lymphoma international prognostic index (A-HIPI) in Turkish patients with classical Hodgkin lymphoma.Ann Hematol. 2025 Mar;104(3):1765-1775. doi: 10.1007/s00277-025-06292-3. Epub 2025 Mar 12. Ann Hematol. 2025. PMID: 40069436 Free PMC article.
-
Prognostic Factors for Clinical Outcomes in Patients with Newly Diagnosed Advanced-stage Hodgkin Lymphoma: A Nationwide Retrospective Study.Cancer Control. 2022 Jan-Dec;29:10732748221124865. doi: 10.1177/10732748221124865. Cancer Control. 2022. PMID: 36134681 Free PMC article.
-
Unilateral Ptosis in an Older Adult; a Rare Presentation of Primary Extranodal Hodgkin's Lymphoma.Case Rep Oncol. 2022 Feb 14;15(1):106-113. doi: 10.1159/000520715. eCollection 2022 Jan-Apr. Case Rep Oncol. 2022. PMID: 35350801 Free PMC article.
-
Prognostic Nutritional Index (PNI) as a potential predictor and intervention target for perioperative ischemic stroke: a retrospective cohort study.BMC Anesthesiol. 2023 Aug 10;23(1):268. doi: 10.1186/s12871-023-02216-8. BMC Anesthesiol. 2023. PMID: 37563630 Free PMC article.
References
-
- Advani RH, Chen H, Habermann TM, Morrison VA, Weller EA, Fisher RI, Peterson BA, Gascoyne RD, Horning SJ Eastern Cooperative Oncology, G.,Cancer, Leukemia Group, B. & Southwest Oncology, G. Comparison of conventional prognostic indices in patients older than 60 years with diffuse large B-cell lymphoma treated with R-CHOP in the US Intergroup Study (ECOG 4494, CALGB 9793): consideration of age greater than 70 years in an elderly prognostic index (E-IPI) Br J Haematol. 2010;151:143–151. - PMC - PubMed
-
- Arai S, Fanale M, DeVos S, Engert A, Illidge T, Borchmann P, Younes A, Morschhauser F, McMillan A, Horning SJ. Defining a Hodgkin lymphoma population for novel therapeutics after relapse from autologous hematopoietic cell transplant. Leuk Lymphoma. 2013;54:2531–2533. - PubMed
-
- Barrington SF, Mikhaeel NG, Kostakoglu L, Meignan M, Hutchings M, Mueller SP, Schwartz LH, Zucca E, Fisher RI, Trotman J, Hoekstra OS, Hicks RJ, O'Doherty MJ, Hustinx R, Biggi A, Cheson BD. Role of imaging in the staging and response assessment of lymphoma: consensus of the International Conference on Malignant Lymphomas Imaging Working Group. J Clin Oncol. 2014;32:3048–3058. - PMC - PubMed
-
- Biggi A, Gallamini A, Chauvie S, Hutchings M, Kostakoglu L, Gregianin M, Meignan M, Malkowski B, Hofman MS, Barrington SF. International validation study for interim PET in ABVD-treated, advanced-stage hodgkin lymphoma: interpretation criteria and concordance rate among reviewers. J Nucl Med. 2013;54:683–690. - PubMed
-
- Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, Lister TA, Alliance AL, Lymphoma G Eastern Cooperative Oncology, G., European Mantle Cell Lymphoma, C., Italian Lymphoma, F., European Organisation for, R., Treatment of Cancer/Dutch Hemato-Oncology, G., Grupo Espanol de Medula, O., German High-Grade Lymphoma Study, G., German Hodgkin's Study, G., Japanese Lymphorra Study, G., Lymphoma Study, A., Group, N.C.T.,Nordic Lymphoma Study, G., Southwest Oncology, G. & United Kingdom National Cancer Research, I. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014;32:3059–3068. - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
- CA21076/CA/NCI NIH HHS/United States
- U10 CA027525/CA/NCI NIH HHS/United States
- CA33601/CA/NCI NIH HHS/United States
- CA77597/CA/NCI NIH HHS/United States
- U10 CA077658/CA/NCI NIH HHS/United States
- U10 CA013650/CA/NCI NIH HHS/United States
- N01 CA032102/CA/NCI NIH HHS/United States
- U10 CA077202/CA/NCI NIH HHS/United States
- CA66636/CA/NCI NIH HHS/United States
- U10 CA021115/CA/NCI NIH HHS/United States
- U10 CA031946/CA/NCI NIH HHS/United States
- U10 CA033601/CA/NCI NIH HHS/United States
- CA180820/CA/NCI NIH HHS/United States
- CA27525/CA/NCI NIH HHS/United States
- CA13650/CA/NCI NIH HHS/United States
- U10 CA077597/CA/NCI NIH HHS/United States
- U10 CA017145/CA/NCI NIH HHS/United States
- U10 CA180846/CA/NCI NIH HHS/United States
- U10 CA021076/CA/NCI NIH HHS/United States
- CA11083/CA/NCI NIH HHS/United States
- U10 CA066636/CA/NCI NIH HHS/United States
- U10 CA077440/CA/NCI NIH HHS/United States
- CA46282/CA/NCI NIH HHS/United States
- U10 CA180850/CA/NCI NIH HHS/United States
- P30 CA060553/CA/NCI NIH HHS/United States
- CA77440/CA/NCI NIH HHS/United States
- CA17145/CA/NCI NIH HHS/United States
- U10 CA032102/CA/NCI NIH HHS/United States
- U10 CA046282/CA/NCI NIH HHS/United States
- U10 CA180790/CA/NCI NIH HHS/United States
- CA77202/CA/NCI NIH HHS/United States
- CA180794/CA/NCI NIH HHS/United States
- U10 CA180820/CA/NCI NIH HHS/United States
- U10 CA023318/CA/NCI NIH HHS/United States
- CA77658/CA/NCI NIH HHS/United States
- CA180790/CA/NCI NIH HHS/United States
- U10 CA011083/CA/NCI NIH HHS/United States
- U10 CA180794/CA/NCI NIH HHS/United States
- CA180816/CA/NCI NIH HHS/United States
- U10 CA180799/CA/NCI NIH HHS/United States
- CA21115/CA/NCI NIH HHS/United States
- U10 CA180888/CA/NCI NIH HHS/United States
- CA23318/CA/NCI NIH HHS/United States
- CA180799/CA/NCI NIH HHS/United States
- CA31946/CA/NCI NIH HHS/United States
- U10 CA180816/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

