Hepatic Mitochondrial Pyruvate Carrier 1 Is Required for Efficient Regulation of Gluconeogenesis and Whole-Body Glucose Homeostasis
- PMID: 26344103
- PMCID: PMC4754674
- DOI: 10.1016/j.cmet.2015.07.027
Hepatic Mitochondrial Pyruvate Carrier 1 Is Required for Efficient Regulation of Gluconeogenesis and Whole-Body Glucose Homeostasis
Abstract
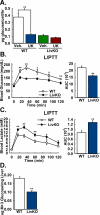
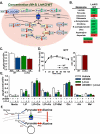
Gluconeogenesis is critical for maintenance of euglycemia during fasting. Elevated gluconeogenesis during type 2 diabetes (T2D) contributes to chronic hyperglycemia. Pyruvate is a major gluconeogenic substrate and requires import into the mitochondrial matrix for channeling into gluconeogenesis. Here, we demonstrate that the mitochondrial pyruvate carrier (MPC) comprising the Mpc1 and Mpc2 proteins is required for efficient regulation of hepatic gluconeogenesis. Liver-specific deletion of Mpc1 abolished hepatic MPC activity and markedly decreased pyruvate-driven gluconeogenesis and TCA cycle flux. Loss of MPC activity induced adaptive utilization of glutamine and increased urea cycle activity. Diet-induced obesity increased hepatic MPC expression and activity. Constitutive Mpc1 deletion attenuated the development of hyperglycemia induced by a high-fat diet. Acute, virally mediated Mpc1 deletion after diet-induced obesity decreased hyperglycemia and improved glucose tolerance. We conclude that the MPC is required for efficient regulation of gluconeogenesis and that the MPC contributes to the elevated gluconeogenesis and hyperglycemia in T2D.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures




Comment in
-
Diabetes: Important role for MPC complex in hepatic gluconeogenesis.Nat Rev Endocrinol. 2015 Nov;11(11):629. doi: 10.1038/nrendo.2015.162. Epub 2015 Sep 22. Nat Rev Endocrinol. 2015. PMID: 26391977 No abstract available.
References
-
- A J, Trygg J, Gullberg J, Johansson AI, Jonsson P, Antti H, Marklund SL, Moritz T. Extraction and GC/MS analysis of the human blood plasma metabolome. Anal Chem. 2005;77:8086–8094. - PubMed
-
- Brivet M, Garcia-Cazorla A, Lyonnet S, Dumez Y, Nassogne MC, Slama A, Boutron A, Touati G, Legrand A, Saudubray JM. Impaired mitochondrial pyruvate importation in a patient and a fetus at risk. Mol Genet Metab. 2003;78:186–192. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R00 AR059190/AR/NIAMS NIH HHS/United States
- R24 DK096518/DK/NIDDK NIH HHS/United States
- R01 DK104998/DK/NIDDK NIH HHS/United States
- R01 DK078184/DK/NIDDK NIH HHS/United States
- T32 HL007638/HL/NHLBI NIH HHS/United States
- T32 HL007121/HL/NHLBI NIH HHS/United States
- P30 CA086862/CA/NCI NIH HHS/United States
- P01 DK058398/DK/NIDDK NIH HHS/United States
- P30CA086862/CA/NCI NIH HHS/United States
- T32 CA078586/CA/NCI NIH HHS/United States
- F32 DK101183/DK/NIDDK NIH HHS/United States
- T32 GM007337/GM/NIGMS NIH HHS/United States
- R01 GM094232/GM/NIGMS NIH HHS/United States
- GM007337/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

