Immunodiagnosis of opisthorchiasis using parasite cathepsin F
- PMID: 26344868
- PMCID: PMC4917378
- DOI: 10.1007/s00436-015-4703-9
Immunodiagnosis of opisthorchiasis using parasite cathepsin F
Abstract
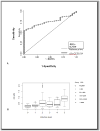
Opisthorchis viverrini, a food-borne trematode parasite endemic in the lower Mekong countries, is conventionally diagnosed by stool examination. However, parasitological stool-based diagnosis can be unreliable in light infections. The goal of this study was to develop the immunodiagnosis of opisthorchiasis using cathepsin F cysteine protease of O. viverrini in both indirect and sandwich ELISA assays. A recombinant O. viverrini cathepsin F (rOv-CF) of 40 kDa was expressed in E. coli strain BL21 (DE3), affinity purified, and deployed in ELISA assays. Human sera from 272 cases were investigated by indirect rOv-CF-based ELISA. Positive antibody response to rOv-CF was found in 137 out of 272 cases (50.37 %) using a cutoff OD (0.400) determined by ROC analysis. In comparison to parasitological stool examined for fluke eggs, the gold standard, the rOv-CF indirect ELISA showed a sensitivity and specificity of 62.1 and 84.05 %, respectively. Serum antibody levels correlated well with egg counts per gram feces (EPG) (P < 0.001). In addition, chicken IgY antibody raised against rOv-CF was tested in a sandwich ELISA for detection of coproantigen in the feces of experimentally infected hamsters. The sandwich ELISA using this chicken IgY in combination with rabbit antibody to O. viverrini somatic antigens showed sensitivity and specificity of 93.3 and 78.57 %, respectively. Together, these findings indicated the potential of rOv-CF for diagnosis of opisthorchiasis, including for uses with chicken IgY for detection of coproantigens of O. viverrini.
Keywords: Cathepsin F; Coproantigen detection; Food-borne trematodiasis; Immunodiagnosis; Opisthorchiasis; Opisthorchis viverrini.
Figures



References
-
- Akai PS, Pungpak S, Chaicumpa W, et al. Serum antibody responses in opisthorchiasis. Int J Parasitol. 1995;25:971–973. - PubMed
-
- Cornelissen JBWJ, Gaasenbeek CPH, Borgsteede FHM, et al. Early immunodiagnosis of fasciolosis in ruminants using recombinant Fasciola hepatica cathepsin L-like protease. Int J Parasitol. 2001;31:728–737. - PubMed
-
- Dixit AK, Dixit P, Sharma RL. Immunodiagnostic-protective role of Cathepsin L cysteine proteinases secreted by Fasciola species. Vet Parasitol. 2008;154:177–184. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

