Lipid-coated gold nanocomposites for enhanced cancer therapy
- PMID: 26345327
- PMCID: PMC4554419
- DOI: 10.2147/IJN.S88307
Lipid-coated gold nanocomposites for enhanced cancer therapy
Abstract
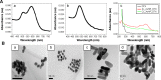
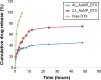
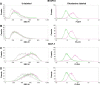
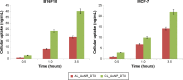
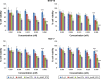
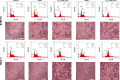
The aim of the work reported here was to develop lipid-coated multifunctional nanocomposites composed of drugs and nanoparticles for use in cancer therapy. We incorporated thermosensitive phospholipids onto the surface of anisotropic gold nanoparticles (AuNPs) to further enhance drug delivery, with possible additional applications for in vivo imaging and photothermal cancer therapy. Lipid-coated nanohybrids loaded with the drug docetaxel (DTX) were prepared by a thin-film formation, hydration, and sonication method. Nanoparticles and their composites were characterized using particle-size analysis, zeta potential measurements, transmission electron microscopy, UV-visible spectroscopy, and reverse-phase high-performance liquid chromatography, demonstrating successful loading of DTX into the lipid bilayer on the surface of the gold nanoparticles. Initial in vitro studies using breast-cancer (MCF-7) and melanoma (B16F10) cell lines demonstrated that the drug-containing nanocomposites at equivalent drug concentrations caused significant cytotoxicity compared to free DTX. Differential flow cytometry analysis confirmed the improved cellular uptake of lipid-coated nanocomposites. Our preliminary results show that DTX-loaded anionic lipid-coated gold nanorod (AL_AuNR_DTX) and cationic lipid-coated gold nanoparticle (CL_AuNP_DTX) possess effective tumor cell-suppression abilities and can therefore be considered promising chemotherapeutic agents. Further evaluation of the therapeutic efficacy of these hybrid nanoparticles combined with external near-infrared photothermal treatment is warranted to assess their synergistic anticancer actions and potential bioimaging applications.
Keywords: anticancer; docetaxel; drug-containing nanocomposites; gold nanorods; thermosensitive lipids.
Figures

Similar articles
-
Plasmonic photothermal release of docetaxel by gold nanoparticles incorporated onto halloysite nanotubes with conjugated 2D8-E3 antibodies for selective cancer therapy.J Nanobiotechnology. 2021 Aug 11;19(1):239. doi: 10.1186/s12951-021-00982-6. J Nanobiotechnology. 2021. PMID: 34380469 Free PMC article.
-
Docetaxel-loaded nanostructured lipid carriers functionalized with trastuzumab (Herceptin) for HER2-positive breast cancer cells.J Liposome Res. 2018 Dec;28(4):285-295. doi: 10.1080/08982104.2017.1370471. Epub 2017 Sep 6. J Liposome Res. 2018. PMID: 28826287
-
The tumor-targeting core-shell structured DTX-loaded PLGA@Au nanoparticles for chemo-photothermal therapy and X-ray imaging.J Control Release. 2015 Dec 28;220(Pt A):545-555. doi: 10.1016/j.jconrel.2015.11.016. Epub 2015 Nov 14. J Control Release. 2015. PMID: 26590021
-
Polymer decorated gold nanoparticles in nanomedicine conjugates.Adv Colloid Interface Sci. 2017 Nov;249:386-399. doi: 10.1016/j.cis.2017.01.007. Epub 2017 Feb 15. Adv Colloid Interface Sci. 2017. PMID: 28259207 Review.
-
Analytical and theranostic applications of gold nanoparticles and multifunctional nanocomposites.Theranostics. 2013;3(3):167-80. doi: 10.7150/thno.5716. Epub 2013 Feb 20. Theranostics. 2013. PMID: 23471188 Free PMC article. Review.
Cited by
-
Understanding the Nano-bio Interfaces: Lipid-Coatings for Inorganic Nanoparticles as Promising Strategy for Biomedical Applications.Front Chem. 2019 May 15;7:343. doi: 10.3389/fchem.2019.00343. eCollection 2019. Front Chem. 2019. PMID: 31165058 Free PMC article. Review.
-
Understanding the near-field photoacoustic spatiotemporal profile from nanostructures.Photoacoustics. 2022 Nov 14;28:100425. doi: 10.1016/j.pacs.2022.100425. eCollection 2022 Dec. Photoacoustics. 2022. PMID: 36425224 Free PMC article.
-
Gold nanoparticle uptake is enhanced by estradiol in MCF-7 breast cancer cells.Int J Nanomedicine. 2019 May 1;14:2705-2718. doi: 10.2147/IJN.S196683. eCollection 2019. Int J Nanomedicine. 2019. PMID: 31118607 Free PMC article.
-
Cholesterol-coated gold nanorods as an efficient nano-carrier for chemotherapeutic delivery and potential treatment of breast cancer: in vitro studies using the MCF-7 cell line.RSC Adv. 2019 Apr 24;9(22):12718-12731. doi: 10.1039/c9ra01041f. eCollection 2019 Apr 17. RSC Adv. 2019. PMID: 35515852 Free PMC article.
-
Reversibly pH-responsive gold nanoparticles and their applications for photothermal cancer therapy.Sci Rep. 2019 Dec 27;9(1):20180. doi: 10.1038/s41598-019-56754-8. Sci Rep. 2019. PMID: 31882911 Free PMC article.
References
-
- Feng SS. New-concept chemotherapy by nanoparticles of biodegradable polymers: where are we now? Nanomedicine (Lond) 2006;1(3):297–309. - PubMed
-
- Muthu MS, Feng SS. Nanopharmacology of liposomes developed for cancer therapy. Nanomedicine (Lond) 2010;5(7):1017–1019. - PubMed
-
- ElBayoumi TA, Torchilin VP. Current trends in liposome research. Methods Mol Biol. 2010;605:1–27. - PubMed
-
- Lasic DD, Papahadjopoulos D. Medical Applications of Liposomes. Amsterdam: Elsevier Science; 1998.
-
- Feng SS, Ruan G, Li QT. Fabrication and characterizations of a novel drug delivery device liposomes-in-microsphere (LIM) Biomaterials. 2004;25(21):5181–5189. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

