Adenylate Cyclase Toxin promotes bacterial internalisation into non phagocytic cells
- PMID: 26346097
- PMCID: PMC4642564
- DOI: 10.1038/srep13774
Adenylate Cyclase Toxin promotes bacterial internalisation into non phagocytic cells
Abstract
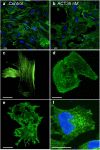
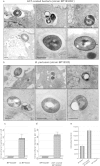
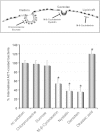
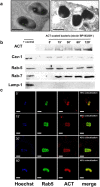
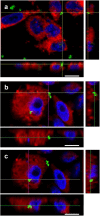
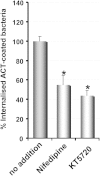
Bordetella pertussis causes whooping cough, a respiratory infectious disease that is the fifth largest cause of vaccine-preventable death in infants. Though historically considered an extracellular pathogen, this bacterium has been detected both in vitro and in vivo inside phagocytic and non-phagocytic cells. However the precise mechanism used by B. pertussis for cell entry, or the putative bacterial factors involved, are not fully elucidated. Here we find that adenylate cyclase toxin (ACT), one of the important toxins of B. pertussis, is sufficient to promote bacterial internalisation into non-phagocytic cells. After characterization of the entry route we show that uptake of "toxin-coated bacteria" proceeds via a clathrin-independent, caveolae-dependent entry pathway, allowing the internalised bacteria to survive within the cells. Intracellular bacteria were found inside non-acidic endosomes with high sphingomyelin and cholesterol content, or "free" in the cytosol of the invaded cells, suggesting that the ACT-induced bacterial uptake may not proceed through formation of late endolysosomes. Activation of Tyr kinases and toxin-induced Ca(2+)-influx are essential for the entry process. We hypothesize that B. pertussis might use ACT to activate the endocytic machinery of non-phagocytic cells and gain entry into these cells, in this way evading the host immune system.
Figures
Similar articles
-
Ca2+ influx and tyrosine kinases trigger Bordetella adenylate cyclase toxin (ACT) endocytosis. Cell physiology and expression of the CD11b/CD18 integrin major determinants of the entry route.PLoS One. 2013 Sep 13;8(9):e74248. doi: 10.1371/journal.pone.0074248. eCollection 2013. PLoS One. 2013. PMID: 24058533 Free PMC article.
-
Selective Enhancement of the Cell-Permeabilizing Activity of Adenylate Cyclase Toxin Does Not Increase Virulence of Bordetella pertussis.Int J Mol Sci. 2021 Oct 28;22(21):11655. doi: 10.3390/ijms222111655. Int J Mol Sci. 2021. PMID: 34769101 Free PMC article.
-
Bordetella filamentous hemagglutinin and adenylate cyclase toxin interactions on the bacterial surface are consistent with FhaB-mediated delivery of ACT to phagocytic cells.mBio. 2024 May 8;15(5):e0063224. doi: 10.1128/mbio.00632-24. Epub 2024 Mar 27. mBio. 2024. PMID: 38534159 Free PMC article.
-
Bioengineering of Bordetella pertussis Adenylate Cyclase Toxin for Vaccine Development and Other Biotechnological Purposes.Toxins (Basel). 2021 Jan 22;13(2):83. doi: 10.3390/toxins13020083. Toxins (Basel). 2021. PMID: 33499260 Free PMC article. Review.
-
Membrane Activity and Channel Formation of the Adenylate Cyclase Toxin (CyaA) of Bordetella pertussis in Lipid Bilayer Membranes.Toxins (Basel). 2020 Mar 10;12(3):169. doi: 10.3390/toxins12030169. Toxins (Basel). 2020. PMID: 32164365 Free PMC article. Review.
Cited by
-
Bordetella Pertussis virulence factors in the continuing evolution of whooping cough vaccines for improved performance.Med Microbiol Immunol. 2018 Feb;207(1):3-26. doi: 10.1007/s00430-017-0524-z. Epub 2017 Nov 21. Med Microbiol Immunol. 2018. PMID: 29164393 Review.
-
Mucosal IgA Antibodies are Critical for Bacterial Clearance of Bordetella pertussis in the Baboon Model.Pathog Immun. 2025 Jun 13;10(2):126-145. doi: 10.20411/pai.v10i2.800. eCollection 2025. Pathog Immun. 2025. PMID: 40612386 Free PMC article.
-
Heterogeneous Family of Cyclomodulins: Smart Weapons That Allow Bacteria to Hijack the Eukaryotic Cell Cycle and Promote Infections.Front Cell Infect Microbiol. 2017 May 23;7:208. doi: 10.3389/fcimb.2017.00208. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28589102 Free PMC article. Review.
-
Caveolae as Potential Hijackable Gates in Cell Communication.Front Cell Dev Biol. 2020 Oct 27;8:581732. doi: 10.3389/fcell.2020.581732. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33195223 Free PMC article. Review.
-
Hepcidin and Ferritin Predict Microbial Etiology in Community-Acquired Pneumonia.Open Forum Infect Dis. 2021 Feb 18;8(4):ofab082. doi: 10.1093/ofid/ofab082. eCollection 2021 Apr. Open Forum Infect Dis. 2021. PMID: 33880390 Free PMC article.
References
-
- Guiso N. Bordetella pertussis: Why is it still circulating? J. Infect. 68, S119–S124 (2014). - PubMed
-
- de Gouw D., Diavatopoulos D. A., Bootsma H. J., Hermans P. W. & Mooi F. R. Pertussis: A matter of immune modulation. FEMS Microbiol. Rev. 35, 441–474 (2011). - PubMed
-
- Higgs R., Higgins S. C., Ross P. J. & Mills K. H. G. Immunity to the respiratory pathogen Bordetella pertussis. Mucosal Immunology 5, 485–500 (2012). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

