A novel bacteriophage cocktail reduces and disperses Pseudomonas aeruginosa biofilms under static and flow conditions
- PMID: 26347362
- PMCID: PMC4720417
- DOI: 10.1111/1751-7915.12316
A novel bacteriophage cocktail reduces and disperses Pseudomonas aeruginosa biofilms under static and flow conditions
Abstract
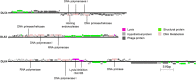
Pseudomonas aeruginosa is an opportunistic human pathogen that forms highly stable communities - biofilms, which contribute to the establishment and maintenance of infections. The biofilm state and intrinsic/acquired bacterial resistance mechanisms contribute to resistance/tolerance to antibiotics that is frequently observed in P. aeruginosa isolates. Here we describe the isolation and characterization of six novel lytic bacteriophages: viruses that infect bacteria, which together efficiently infect and kill a wide range of P. aeruginosa clinical isolates. The phages were used to formulate a cocktail with the potential to eliminate P. aeruginosa PAO1 planktonic cultures. Two biofilm models were studied, one static and one dynamic, and the phage cocktail was assessed for its ability to reduce and disperse the biofilm biomass. For the static model, after 4 h of contact with the phage suspension (MOI 10) more than 95% of biofilm biomass was eliminated. In the flow biofilm model, a slower rate of activity by the phage was observed, but 48 h after addition of the phage cocktail the biofilm was dispersed, with most cells eliminated (> 4 logs) comparing with the control. This cocktail has the potential for development as a therapeutic to control P. aeruginosa infections, which are predominantly biofilm centred.
© 2015 The Authors. Microbial Biotechnology published by John Wiley & Sons Ltd and Society for Applied Microbiology.
Figures



 ), with single
), with single  ),
),  ),
),  ),
),  ),
),  )
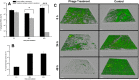
)  ) and with the phage cocktail in
) and with the phage cocktail in  ) and also a negative‐control
) and also a negative‐control  ) are shown in the figure. Assays were performed three times, and
) are shown in the figure. Assays were performed three times, and 
References
-
- Ackermann, H.W. (2006) Classification of Bacteriophages. New York, NY, USA: Oxford University Press.
-
- Adams, M. (1959) Bacteriophages. New York, NY, USA: Interscience Publishers.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

