Efficacy and Safety of Artesunate-Amodiaquine versus Artemether-Lumefantrine in the Treatment of Uncomplicated Plasmodium falciparum Malaria in Sentinel Sites across Côte d'Ivoire
- PMID: 26347849
- PMCID: PMC4549615
- DOI: 10.1155/2015/878132
Efficacy and Safety of Artesunate-Amodiaquine versus Artemether-Lumefantrine in the Treatment of Uncomplicated Plasmodium falciparum Malaria in Sentinel Sites across Côte d'Ivoire
Abstract
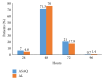
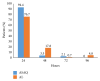
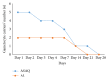
Two years after the introduction of free Artesunate-Amodiaquine (ASAQ) and Artemether-Lumefantrine (AL) for the treatment of uncomplicated malaria in public health facilities in Côte d'Ivoire, we carried out this study to compare their efficacy and tolerability in three surveillance sites. It was a multicentre open randomised clinical trial of 3-day ASAQ treatment against AL for the treatment of 2 parallel groups of patients aged 2 years and above. The endpoints were (1) Adequate Clinical and Parasitological Response (ACPR) at day 28 and (2) the clinical and biological tolerability. Of the 300 patients who were enrolled 289, with 143 (49.5%) and 146 (50.5%) in the ASAQ and AL groups, respectively, correctly followed the WHO 2003 protocol we used. The PCR-corrected ACPR was 99.3% for each group. More than 94% of patients no longer showed signs of fever, 48 hours after treatment. Approximately 78% of the people in the ASAQ group had a parasite clearance time of 48 hours or less compared to 81% in the AL group (p = 0.496). Both drugs were found to be well tolerated by the patients. This study demonstrates the effectiveness and tolerability of ASAQ and AL supporting their continuous use for the treatment of uncomplicated P. falciparum malaria infection in Côte d'Ivoire.
Figures
Similar articles
-
Meta-Analysis of Data from Four Clinical Trials in the Ivory Coast Assessing the Efficacy of Two Artemisinin-Based Combination Therapies (Artesunate-Amodiaquine and Artemether-Lumefantrine) between 2009 and 2016.Trop Med Infect Dis. 2023 Dec 29;9(1):10. doi: 10.3390/tropicalmed9010010. Trop Med Infect Dis. 2023. PMID: 38251206 Free PMC article.
-
Efficacy and tolerability of artesunate-amodiaquine versus artemether-lumefantrine in the treatment of uncomplicated Plasmodium falciparum malaria at two sentinel sites across Côte d’Ivore.Ann Parasitol. 2018;64(1):49–57. doi: 10.17420/ap6401.132. Ann Parasitol. 2018. PMID: 29717574 Clinical Trial.
-
Efficacy and safety of artesuante-amodiaquine and artemether lumefantrine, the first line malaria treatment in six sentinel's sites of Côte d'Ivoire, West Africa.Ann Parasitol. 2020;66(4):561-571. doi: 10.17420/ap6604.299. Ann Parasitol. 2020. PMID: 33789028 Clinical Trial.
-
Efficacy and tolerability of artesunate-amodiaquine (Camoquin plus) versus artemether-lumefantrine (Coartem) against uncomplicated Plasmodium falciparum malaria: multisite trial in Senegal and Ivory Coast.Trop Med Int Health. 2010 May;15(5):608-13. doi: 10.1111/j.1365-3156.2010.02487.x. Epub 2010 Mar 1. Trop Med Int Health. 2010. PMID: 20214761 Clinical Trial.
-
Therapeutic efficacy and safety of artesunate + amodiaquine and artemether + lumefantrine in treating uncomplicated Plasmodium falciparum malaria in children on the rainy south-east coast of Madagascar.Parasite. 2023;30:32. doi: 10.1051/parasite/2023034. Epub 2023 Aug 30. Parasite. 2023. PMID: 37646608 Free PMC article. Clinical Trial.
Cited by
-
Therapeutic efficacy of artemether-lumefantrine, artesunate-amodiaquine and dihydroartemisinin-piperaquine in the treatment of uncomplicated Plasmodium falciparum malaria in Sub-Saharan Africa: A systematic review and meta-analysis.PLoS One. 2022 Mar 10;17(3):e0264339. doi: 10.1371/journal.pone.0264339. eCollection 2022. PLoS One. 2022. PMID: 35271592 Free PMC article.
-
Meta-Analysis of Data from Four Clinical Trials in the Ivory Coast Assessing the Efficacy of Two Artemisinin-Based Combination Therapies (Artesunate-Amodiaquine and Artemether-Lumefantrine) between 2009 and 2016.Trop Med Infect Dis. 2023 Dec 29;9(1):10. doi: 10.3390/tropicalmed9010010. Trop Med Infect Dis. 2023. PMID: 38251206 Free PMC article.
-
Increase of Plasmodium falciparum parasites carrying lumefantrine-tolerance molecular markers and lack of South East Asian pfk13 artemisinin-resistance mutations in samples collected from 2013 to 2016 in Côte d'Ivoire.J Parasit Dis. 2024 Mar;48(1):59-66. doi: 10.1007/s12639-023-01640-4. Epub 2024 Jan 12. J Parasit Dis. 2024. PMID: 38440764 Free PMC article.
-
Effectiveness and safety of artesunate-amodiaquine versus artemether-lumefantrine for home-based treatment of uncomplicated Plasmodium falciparum malaria among children 6-120 months in Yaoundé, Cameroon: a randomized trial.BMC Infect Dis. 2022 Feb 21;22(1):166. doi: 10.1186/s12879-022-07101-2. BMC Infect Dis. 2022. PMID: 35189818 Free PMC article. Clinical Trial.
-
Malaria parasite clearance from patients following artemisinin-based combination therapy in Côte d'Ivoire.Infect Drug Resist. 2018 Oct 26;11:2031-2038. doi: 10.2147/IDR.S167518. eCollection 2018. Infect Drug Resist. 2018. PMID: 30464545 Free PMC article.
References
-
- World Health Organization. World Malaria Report. Geneva, Switzerland: World Health Organization; 2013.
-
- Ministère de la Santé et de l'Hygiène Publique. PNLP. Directives de prise en charge du paludisme. Abidjan, Côte d'Ivoire: PNLP; 2008.
-
- Brice B. K., William Y., Lacina O., et al. In vitro susceptibility of Plasmodium falciparum isolates from Abidjan, Côte d'Ivoire, to artemisinin, chloroquine, dihydroartemisinin and pyronaridine. Tanzania Journal of Health Research. 2010;12(supplement 1):73–79. - PubMed
-
- World Health Organization. Antimalarial Drug Combination Therapy: Report of a Technical Consultation. Geneva, Switzerland: World Health Organization; 2001.
-
- Ministère de la santé et de l’hygiène publique. Arrêté N°024/CAB/MSHP du 02 janvier 2007 portant institution d’un schéma thérapeutique pour le traitement du paludisme en Côte d’Ivoire. Abidjan: Journal officiel, 2007.
LinkOut - more resources
Full Text Sources
Other Literature Sources