Epigenetic Regulation of Antibody Responses by the Histone H2A Deubiquitinase MYSM1
- PMID: 26348977
- PMCID: PMC4562257
- DOI: 10.1038/srep13755
Epigenetic Regulation of Antibody Responses by the Histone H2A Deubiquitinase MYSM1
Abstract
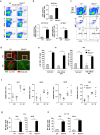
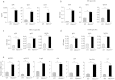
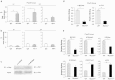
B cell-mediated antibody response plays critical roles in protective immunity, as well as in the pathogenesis of allergic and autoimmune diseases. Epigenetic histone and DNA modifications regulate gene transcription and immunity; however, so far, little is known about the role of epigenetic regulation in antibody responses. In this study, we found that mice deficient in the histone H2A deubiquitinase MYSM1, despite their severe defect in B cell development, exhibit an enhanced antibody response against both T cell-dependent and independent antigens. We revealed that MYSM1 intrinsically represses plasma cell differentiation and antibody production. Mechanistic studies demonstrated that MYSM1 is a transcriptional activator of Pax5, the repressors of plasma cell differentiation, by facilitating key transcriptional factor recruitment and coordinating histone modifications at the Pax5 loci. Hence, this study uncovers a critical role for MYSM1 in epigenetically repressing plasma cell differentiation and antibody production, in addition to its opposing, active role in B cell development. Importantly, this study further provides a new target and strategy to modulate antibody production and responses with profound therapeutic implications.
Figures




References
-
- Kouzarides T. Chromatin modifications and their function. Cell 128, 693–705 (2007). - PubMed
-
- Li B., Carey M. & Workman J. L. The role of chromatin during transcription. Cell 128, 707–719 (2007). - PubMed
-
- Jenuwein T. & Allis C. D. Translating the histone code. Science 293, 1074–1080 (2001). - PubMed
-
- Strahl B. D. & Allis C. D. The language of covalent histone modifications. Nature 403, 41–45 (2000). - PubMed
-
- Suganuma T. & Workman J. L. Signals and combinatorial functions of histone modifications. Annu. Rev. Biochem. 80, 473–499 (2011). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

