CD4(+) T-cell survival in the GI tract requires dectin-1 during fungal infection
- PMID: 26349660
- PMCID: PMC4677461
- DOI: 10.1038/mi.2015.79
CD4(+) T-cell survival in the GI tract requires dectin-1 during fungal infection
Abstract
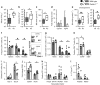
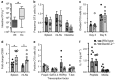
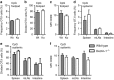
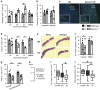
Dectin-1 is an innate antifungal C-type lectin receptor necessary for protective antifungal immunity. We recently discovered that Dectin-1 is involved in controlling fungal infections of the gastrointestinal (GI) tract, but how this C-type lectin receptor mediates these activities is unknown. Here, we show that Dectin-1 is essential for driving fungal-specific CD4(+) T-cell responses in the GI tract. Loss of Dectin-1 resulted in abrogated dendritic cell responses in the mesenteric lymph nodes (mLNs) and defective T-cell co-stimulation, causing substantial increases in CD4(+) T-cell apoptosis and reductions in the cellularity of GI-associated lymphoid tissues. CD8(+) T-cell responses were unaffected by Dectin-1 deficiency. These functions of Dectin-1 have significant implications for our understanding of intestinal immunity and susceptibility to fungal infections.
Figures


Similar articles
-
Altered CD4+ T cell phenotype and function determine the susceptibility to mucosal candidiasis in transgenic mice expressing HIV-1.J Immunol. 2006 Jul 1;177(1):479-91. doi: 10.4049/jimmunol.177.1.479. J Immunol. 2006. PMID: 16785545
-
The β-glucan receptor Dectin-1 activates the integrin Mac-1 in neutrophils via Vav protein signaling to promote Candida albicans clearance.Cell Host Microbe. 2011 Dec 15;10(6):603-15. doi: 10.1016/j.chom.2011.10.009. Cell Host Microbe. 2011. PMID: 22177564 Free PMC article.
-
The protective effect of inflammatory monocytes during systemic C. albicans infection is dependent on collaboration between C-type lectin-like receptors.PLoS Pathog. 2019 Jun 26;15(6):e1007850. doi: 10.1371/journal.ppat.1007850. eCollection 2019 Jun. PLoS Pathog. 2019. PMID: 31242262 Free PMC article.
-
Dectin-1 is not required for controlling Candida albicans colonization of the gastrointestinal tract.Infect Immun. 2012 Dec;80(12):4216-22. doi: 10.1128/IAI.00559-12. Epub 2012 Sep 17. Infect Immun. 2012. PMID: 22988015 Free PMC article.
-
Recognition of non-self-polysaccharides by C-type lectin receptors dectin-1 and dectin-2.Glycobiology. 2009 Jun;19(6):568-75. doi: 10.1093/glycob/cwp032. Epub 2009 Mar 14. Glycobiology. 2009. PMID: 19287024 Free PMC article. Review.
Cited by
-
Similar proteome expression profiles of the aggregated lymphoid nodules area and Peyer's patches in Bactrian camel.BMC Genomics. 2023 Oct 11;24(1):608. doi: 10.1186/s12864-023-09715-5. BMC Genomics. 2023. PMID: 37821839 Free PMC article.
-
The Dectin-1 and Dectin-2 clusters: C-type lectin receptors with fundamental roles in immunity.EMBO Rep. 2024 Dec;25(12):5239-5264. doi: 10.1038/s44319-024-00296-2. Epub 2024 Oct 31. EMBO Rep. 2024. PMID: 39482490 Free PMC article. Review.
-
Insights on the Functional Role of Beta-Glucans in Fungal Immunity Using Receptor-Deficient Mouse Models.Int J Mol Sci. 2021 Apr 30;22(9):4778. doi: 10.3390/ijms22094778. Int J Mol Sci. 2021. PMID: 33946381 Free PMC article. Review.
-
Immune Interactions with Pathogenic and Commensal Fungi: A Two-Way Street.Immunity. 2015 Nov 17;43(5):845-58. doi: 10.1016/j.immuni.2015.10.023. Immunity. 2015. PMID: 26588778 Free PMC article. Review.
-
Immune Sensing and Potential Immunotherapeutic Approaches to Control Chromoblastomycosis.J Fungi (Basel). 2020 Dec 22;7(1):3. doi: 10.3390/jof7010003. J Fungi (Basel). 2020. PMID: 33375204 Free PMC article. Review.
References
-
- Saijo, S. et al. Dectin-1 is required for host defense against Pneumocystis carinii but not against Candida albicans. Nat. Immunol. 8, 39–46 (2007). - PubMed
-
- LeibundGut-Landmann, S. et al. Syk- and CARD9-dependent coupling of innate immunity to the induction of T helper cells that produce interleukin 17. Nat. Immunol. 8, 630–638 (2007). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

