Baicalin inhibits the lethality of Shiga-like toxin 2 in mice
- PMID: 26349825
- PMCID: PMC4604387
- DOI: 10.1128/AAC.01416-15
Baicalin inhibits the lethality of Shiga-like toxin 2 in mice
Abstract
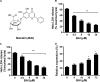
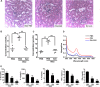
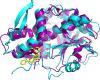
Shiga-like toxins (Stxs), produced by pathogenic Escherichia coli, are a major virulence factor involved in severe diseases in human and animals. These toxins are ribosome-inactivating proteins, and treatment for diseases caused by them is not available. Therefore, there is an urgent need for agents capable of effectively targeting this lethal toxin. In this study, we identified baicalin, a flavonoid compound used in Chinese traditional medicine, as a compound against Shiga-like toxin 2 (Stx2). We found that baicalin significantly improves renal function and reduces Stx2-induced lethality in mice. Further experiments revealed that baicalin induces the formation of oligomers by the toxin by direct binding. We also identified the residues important for such interactions and analyzed their roles in binding baicalin by biophysical and biochemical analyses. Our results establish baicalin as a candidate compound for the development of therapeutics against diseases caused by Stxs.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures
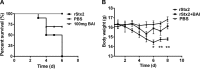

Similar articles
-
A potential therapeutic peptide-based neutralizer that potently inhibits Shiga toxin 2 in vitro and in vivo.Sci Rep. 2016 Feb 23;6:21837. doi: 10.1038/srep21837. Sci Rep. 2016. PMID: 26903273 Free PMC article.
-
Baicalin Protects Mice from Lethal Infection by Enterohemorrhagic Escherichia coli.Front Microbiol. 2017 Mar 9;8:395. doi: 10.3389/fmicb.2017.00395. eCollection 2017. Front Microbiol. 2017. PMID: 28337193 Free PMC article.
-
The serine 31 residue of the B subunit of Shiga toxin 2 is essential for secretion in enterohemorrhagic Escherichia coli.Infect Immun. 2007 May;75(5):2189-200. doi: 10.1128/IAI.01546-06. Epub 2007 Feb 26. Infect Immun. 2007. PMID: 17325057 Free PMC article.
-
Exosome-associated Shiga toxin 2 is released from cells and causes severe toxicity in mice.Sci Rep. 2018 Jul 17;8(1):10776. doi: 10.1038/s41598-018-29128-9. Sci Rep. 2018. PMID: 30018364 Free PMC article.
-
Escherichia coli serogroup O107/O117 lipopolysaccharide binds and neutralizes Shiga toxin 2.J Bacteriol. 2004 Aug;186(16):5506-12. doi: 10.1128/JB.186.16.5506-5512.2004. J Bacteriol. 2004. PMID: 15292153 Free PMC article.
Cited by
-
Reversal of Azithromycin Resistance in Staphylococcus saprophyticus by Baicalin.Front Vet Sci. 2022 Feb 18;9:827674. doi: 10.3389/fvets.2022.827674. eCollection 2022. Front Vet Sci. 2022. PMID: 35252422 Free PMC article.
-
Antibacterial Flavonoids from Medicinal Plants Covalently Inactivate Type III Protein Secretion Substrates.J Am Chem Soc. 2016 Feb 24;138(7):2209-18. doi: 10.1021/jacs.5b11575. Epub 2016 Feb 16. J Am Chem Soc. 2016. PMID: 26847396 Free PMC article.
-
Phytochemicals Controlling Enterohemorrhagic Escherichia coli (EHEC) Virulence-Current Knowledge of Their Mechanisms of Action.Int J Mol Sci. 2025 Jan 4;26(1):381. doi: 10.3390/ijms26010381. Int J Mol Sci. 2025. PMID: 39796236 Free PMC article. Review.
-
Effect of Baicalin-Aluminum Complexes on Fecal Microbiome in Piglets.Int J Mol Sci. 2019 May 14;20(10):2390. doi: 10.3390/ijms20102390. Int J Mol Sci. 2019. PMID: 31091773 Free PMC article.
-
Plant-Derived Products with Therapeutic Potential against Gastrointestinal Bacteria.Pathogens. 2023 Feb 15;12(2):333. doi: 10.3390/pathogens12020333. Pathogens. 2023. PMID: 36839605 Free PMC article. Review.
References
-
- Griffin PM, Tauxe RV. 1991. The epidemiology of infections caused by Escherichia coli O157:H7, other enterohemorrhagic E. coli, and the associated hemolytic uremic syndrome. Epidemiol Rev 13:60–98. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

