A Genetic Selection for dinB Mutants Reveals an Interaction between DNA Polymerase IV and the Replicative Polymerase That Is Required for Translesion Synthesis
- PMID: 26352807
- PMCID: PMC4564189
- DOI: 10.1371/journal.pgen.1005507
A Genetic Selection for dinB Mutants Reveals an Interaction between DNA Polymerase IV and the Replicative Polymerase That Is Required for Translesion Synthesis
Abstract
Translesion DNA synthesis (TLS) by specialized DNA polymerases (Pols) is a conserved mechanism for tolerating replication blocking DNA lesions. The actions of TLS Pols are managed in part by ring-shaped sliding clamp proteins. In addition to catalyzing TLS, altered expression of TLS Pols impedes cellular growth. The goal of this study was to define the relationship between the physiological function of Escherichia coli Pol IV in TLS and its ability to impede growth when overproduced. To this end, 13 novel Pol IV mutants were identified that failed to impede growth. Subsequent analysis of these mutants suggest that overproduced levels of Pol IV inhibit E. coli growth by gaining inappropriate access to the replication fork via a Pol III-Pol IV switch that is mechanistically similar to that used under physiological conditions to coordinate Pol IV-catalyzed TLS with Pol III-catalyzed replication. Detailed analysis of one mutant, Pol IV-T120P, and two previously described Pol IV mutants impaired for interaction with either the rim (Pol IVR) or the cleft (Pol IVC) of the β sliding clamp revealed novel insights into the mechanism of the Pol III-Pol IV switch. Specifically, Pol IV-T120P retained complete catalytic activity in vitro but, like Pol IVR and Pol IVC, failed to support Pol IV TLS function in vivo. Notably, the T120P mutation abrogated a biochemical interaction of Pol IV with Pol III that was required for Pol III-Pol IV switching. Taken together, these results support a model in which Pol III-Pol IV switching involves interaction of Pol IV with Pol III, as well as the β clamp rim and cleft. Moreover, they provide strong support for the view that Pol III-Pol IV switching represents a vitally important mechanism for regulating TLS in vivo by managing access of Pol IV to the DNA.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
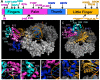

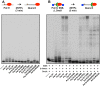
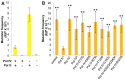

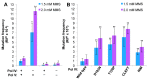
Similar articles
-
During Translesion Synthesis, Escherichia coli DinB89 (T120P) Alters Interactions of DinB (Pol IV) with Pol III Subunit Assemblies and SSB, but Not with the β Clamp.J Bacteriol. 2022 Apr 19;204(4):e0061121. doi: 10.1128/jb.00611-21. Epub 2022 Mar 14. J Bacteriol. 2022. PMID: 35285726 Free PMC article.
-
A bipartite interaction with the processivity clamp potentiates Pol IV-mediated TLS.Proc Natl Acad Sci U S A. 2025 Mar 4;122(9):e2421471122. doi: 10.1073/pnas.2421471122. Epub 2025 Feb 24. Proc Natl Acad Sci U S A. 2025. PMID: 39993197 Free PMC article.
-
Escherichia coli DNA polymerase IV (Pol IV), but not Pol II, dynamically switches with a stalled Pol III* replicase.J Bacteriol. 2012 Jul;194(14):3589-600. doi: 10.1128/JB.00520-12. Epub 2012 Apr 27. J Bacteriol. 2012. PMID: 22544274 Free PMC article.
-
A Comprehensive View of Translesion Synthesis in Escherichia coli.Microbiol Mol Biol Rev. 2020 Jun 17;84(3):e00002-20. doi: 10.1128/MMBR.00002-20. Print 2020 Aug 19. Microbiol Mol Biol Rev. 2020. PMID: 32554755 Free PMC article. Review.
-
Translesion synthesis in Escherichia coli: lessons from the NarI mutation hot spot.DNA Repair (Amst). 2007 Jul 1;6(7):1032-41. doi: 10.1016/j.dnarep.2007.02.021. Epub 2007 Apr 2. DNA Repair (Amst). 2007. PMID: 17403618 Review.
Cited by
-
A gatekeeping function of the replicative polymerase controls pathway choice in the resolution of lesion-stalled replisomes.Proc Natl Acad Sci U S A. 2019 Dec 17;116(51):25591-25601. doi: 10.1073/pnas.1914485116. Epub 2019 Dec 3. Proc Natl Acad Sci U S A. 2019. PMID: 31796591 Free PMC article.
-
DNA Polymerases ImuC and DinB Are Involved in DNA Alkylation Damage Tolerance in Pseudomonas aeruginosa and Pseudomonas putida.PLoS One. 2017 Jan 24;12(1):e0170719. doi: 10.1371/journal.pone.0170719. eCollection 2017. PLoS One. 2017. PMID: 28118378 Free PMC article.
-
A bipartite interaction with the processivity clamp potentiates Pol IV-mediated TLS.bioRxiv [Preprint]. 2024 May 31:2024.05.30.596738. doi: 10.1101/2024.05.30.596738. bioRxiv. 2024. Update in: Proc Natl Acad Sci U S A. 2025 Mar 04;122(9):e2421471122. doi: 10.1073/pnas.2421471122. PMID: 38853898 Free PMC article. Updated. Preprint.
-
Observing protein dynamics during DNA-lesion bypass by the replisome.Front Mol Biosci. 2022 Sep 21;9:968424. doi: 10.3389/fmolb.2022.968424. eCollection 2022. Front Mol Biosci. 2022. PMID: 36213113 Free PMC article. Review.
-
DNA Polymerase III, but Not Polymerase IV, Must Be Bound to a τ-Containing DnaX Complex to Enable Exchange into Replication Forks.J Biol Chem. 2016 May 27;291(22):11727-35. doi: 10.1074/jbc.M116.725358. Epub 2016 Apr 7. J Biol Chem. 2016. PMID: 27056333 Free PMC article.
References
-
- Friedberg EC, Walker GC, Siede W, Wood RD, Schultz RA, et al. (2006) DNA repair and mutagenesis Washington, D. C.: ASM Press.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

