Identification of tumour-reactive lymphatic endothelial cells capable of inducing progression of gastric cancer
- PMID: 26355233
- PMCID: PMC4651131
- DOI: 10.1038/bjc.2015.282
Identification of tumour-reactive lymphatic endothelial cells capable of inducing progression of gastric cancer
Abstract
Background: Tumour cells and stromal cells interact in the tumour microenvironment; moreover, stromal cells can acquire abnormalities that contribute to tumour progression. However, interactions between lymphatic endothelial cells (LECs) and tumour cells are largely unexamined. In this study, we aimed to determine whether tumour-specific LECs inhabit the tumour microenvironment and examine their influence on this microenvironment.
Methods: We isolated normal LECs (NLECs) from a non-metastatic lymph node and tumour-associated LECs (TLECs) from cancerous lymph nodes. We examined proliferative and migratory potency, growth factor production, and gene expression of each type of LEC. Moreover, we developed a co-culture system to investigate the interactions between gastric cancer cells and LECs.
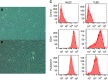
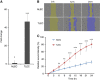
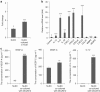
Results: When compared with NLEC, TLECs had an abnormal shape, high proliferative and migratory abilities, and elevated expression of genes associated with inflammation, cell growth, and cell migration. NLECs co-cultured with gastric cancer cells from the OCUM12 cell line acquired TLEC-like phenotypes. Also, OCUM12 cells co-cultured with TLECs expressed high levels of genes responsible for metastasis.
Conclusions: Our results demonstrated that LECs interacted with tumour cells and obtained abnormal phenotypes that could have important roles in tumour progression.
Figures



Similar articles
-
Immunoregulatory Function of Lymphatic Endothelial Cells in Tumor-draining Lymph Nodes of Human Gastric Cancer.Anticancer Res. 2017 Jun;37(6):2875-2883. doi: 10.21873/anticanres.11640. Anticancer Res. 2017. PMID: 28551624
-
Lymphatic endothelial cell-secreted CXCL1 stimulates lymphangiogenesis and metastasis of gastric cancer.Int J Cancer. 2012 Feb 15;130(4):787-97. doi: 10.1002/ijc.26035. Epub 2011 May 9. Int J Cancer. 2012. PMID: 21387301
-
Tumor-associated macrophages induce capillary morphogenesis of lymphatic endothelial cells derived from human gastric cancer.Cancer Sci. 2016 Aug;107(8):1101-9. doi: 10.1111/cas.12977. Epub 2016 Jun 22. Cancer Sci. 2016. PMID: 27227358 Free PMC article.
-
Single-Cell RNA Sequencing Reveals Heterogeneity and Functional Diversity of Lymphatic Endothelial Cells.Int J Mol Sci. 2021 Nov 5;22(21):11976. doi: 10.3390/ijms222111976. Int J Mol Sci. 2021. PMID: 34769408 Free PMC article. Review.
-
Lymph Node Lymphatic Endothelial Cell Expansion and Contraction and the Programming of the Immune Response.Front Immunol. 2019 Jan 25;10:36. doi: 10.3389/fimmu.2019.00036. eCollection 2019. Front Immunol. 2019. PMID: 30740101 Free PMC article. Review.
Cited by
-
Effect of crosstalk among conspirators in tumor microenvironment on niche metastasis of gastric cancer.Am J Cancer Res. 2022 Dec 15;12(12):5375-5402. eCollection 2022. Am J Cancer Res. 2022. PMID: 36628284 Free PMC article. Review.
-
Integrated profiling identifies SLC5A6 and MFAP2 as novel diagnostic and prognostic biomarkers in gastric cancer patients.Int J Oncol. 2020 Feb;56(2):460-469. doi: 10.3892/ijo.2019.4944. Epub 2019 Dec 16. Int J Oncol. 2020. PMID: 31894266 Free PMC article.
-
Endothelial Cells Promote Colorectal Cancer Cell Survival by Activating the HER3-AKT Pathway in a Paracrine Fashion.Mol Cancer Res. 2019 Jan;17(1):20-29. doi: 10.1158/1541-7786.MCR-18-0341. Epub 2018 Aug 21. Mol Cancer Res. 2019. PMID: 30131447 Free PMC article.
-
Tumor microenvironment in gastric cancers.Cancer Sci. 2020 Aug;111(8):2696-2707. doi: 10.1111/cas.14521. Epub 2020 Jul 11. Cancer Sci. 2020. PMID: 32519436 Free PMC article. Review.
-
Targeting the tumor stroma for cancer therapy.Mol Cancer. 2022 Nov 2;21(1):208. doi: 10.1186/s12943-022-01670-1. Mol Cancer. 2022. PMID: 36324128 Free PMC article. Review.
References
-
- Apte RN, Krelin Y, Song X, Dotan S, Recih E, Elkabets M, Carmi Y, Dvorkin T, White RM, Gayvoronsky L, Segal S, Voronov E (2006) Effects of micro-environment- and malignant cell-derived interleukin-1 in carcinogenesis, tumour invasiveness and tumour-host interactions. Eur J Cancer 42(6): 751–759. - PubMed
-
- Bollrath J, Phesse TJ, von Burstin VA, Putoczki T, Bennecke M, Bateman T, Nebelsiek T, Lundgren-May T, Canli O, Schwitalla S, Matthews V, Schmid RM, Kirchner T, Arkan MC, Ernst M, Greten FR (2009) gp130-mediated Stat3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell 15(2): 91–102. - PubMed
-
- Fridlender ZG, Albelda SM (2012) Tumor-associated neutrophils: friend or foe? Carcinogenesis 33(5): 949–955. - PubMed
-
- Giavazzi R, Garofalo A, Bani MR, Abbate M, Ghezzi P, Boraschi D, Mantovani A, Dejana E (1990) Interleukin 1-induced augmentation of experimental metastases from a human melanoma in nude mice. Cancer Res 50(15): 4771–4775. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

