Galectins regulate the inflammatory response in airway epithelial cells exposed to microbial neuraminidase by modulating the expression of SOCS1 and RIG1
- PMID: 26355912
- PMCID: PMC4624043
- DOI: 10.1016/j.molimm.2015.08.005
Galectins regulate the inflammatory response in airway epithelial cells exposed to microbial neuraminidase by modulating the expression of SOCS1 and RIG1
Abstract
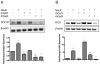
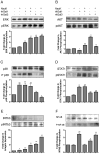
Influenza patients frequently display increased susceptibility to Streptococcus pneumoniae co-infection and sepsis, the prevalent cause of mortality during influenza pandemics. However, the detailed mechanisms by which an influenza infection predisposes patients to suffer pneumococcal pneumonia are not fully understood. A murine model for influenza infection closely reflects the observations in human patients, since if the animals that have recovered from influenza A virus (IAV) sublethal infection are challenged with S. pneumoniae, they undergo a usually fatal uncontrolled cytokine response. We have previously demonstrated both in vitro and in vivo that the expression and secretion of galectin-1 (Gal1) and galectin-3 (Gal3) are modulated during IAV infection, and that the viral neuraminidase unmasks galactosyl moieties in the airway epithelia. In this study we demonstrate in vitro that the binding of secreted Gal1 and Gal3 to the epithelial cell surface modulates the expression of SOCS1 and RIG1, and activation of ERK, AKT or JAK/STAT1 signaling pathways, leading to a disregulated expression and release of pro-inflammatory cytokines. Our results suggest that the activity of the viral and pneumococcal neuraminidases on the surface of the airway epithelial cells function as a "danger signal" that leads to rapid upregulation of SOCS1 expression to prevent an uncontrolled inflammatory response. The binding of extracellular Gal1 or Gal3 to the galactosyl moieties unmasked on the surface of airway epithelial cells can either "fine-tune" or severely disregulate this process, respectively, the latter potentially leading to hypercytokinemia.
Keywords: Chemokines; Cytokines; Galectin; Neuraminidase; RIG1; SOCS; Signaling pathway.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Desialylation of airway epithelial cells during influenza virus infection enhances pneumococcal adhesion via galectin binding.Mol Immunol. 2015 May;65(1):1-16. doi: 10.1016/j.molimm.2014.12.010. Epub 2015 Jan 16. Mol Immunol. 2015. PMID: 25597246 Free PMC article.
-
Galectin-3 disrupts tight junctions of airway epithelial cell monolayers by inducing expression and release of matrix metalloproteinases upon influenza A infection.Glycobiology. 2025 Jan 13;35(1):cwae093. doi: 10.1093/glycob/cwae093. Glycobiology. 2025. PMID: 39569730
-
Cutting edge: innate immune response triggered by influenza A virus is negatively regulated by SOCS1 and SOCS3 through a RIG-I/IFNAR1-dependent pathway.J Immunol. 2008 Feb 15;180(4):2034-8. doi: 10.4049/jimmunol.180.4.2034. J Immunol. 2008. PMID: 18250407
-
SOCS proteins: negative regulators of cytokine signaling.Stem Cells. 2001;19(5):378-87. doi: 10.1634/stemcells.19-5-378. Stem Cells. 2001. PMID: 11553846 Review.
-
SOCS3 and its role in associated diseases.Hum Immunol. 2015 Oct;76(10):775-80. doi: 10.1016/j.humimm.2015.09.037. Epub 2015 Sep 30. Hum Immunol. 2015. PMID: 26429311 Review.
Cited by
-
The Antiviral Role of Galectins toward Influenza A Virus Infection-An Alternative Strategy for Influenza Therapy.Pharmaceuticals (Basel). 2021 May 20;14(5):490. doi: 10.3390/ph14050490. Pharmaceuticals (Basel). 2021. PMID: 34065500 Free PMC article. Review.
-
Galectin-1 mediates interactions between polymorphonuclear leukocytes and vascular endothelial cells, and promotes their extravasation during lipopolysaccharide-induced acute lung injury.Mol Immunol. 2023 Apr;156:127-135. doi: 10.1016/j.molimm.2023.02.011. Epub 2023 Mar 13. Mol Immunol. 2023. PMID: 36921487 Free PMC article.
-
Glycosylation-dependent galectin-receptor interactions promote Chlamydia trachomatis infection.Proc Natl Acad Sci U S A. 2018 Jun 26;115(26):E6000-E6009. doi: 10.1073/pnas.1802188115. Epub 2018 Jun 11. Proc Natl Acad Sci U S A. 2018. PMID: 29891717 Free PMC article.
-
Influenza Virus Neuraminidase Engages CD83 and Promotes Pulmonary Injury.J Virol. 2021 Jan 13;95(3):e01753-20. doi: 10.1128/JVI.01753-20. Print 2021 Jan 13. J Virol. 2021. PMID: 33177200 Free PMC article.
-
Galectin-3 inhibitor GB0139 protects against acute lung injury by inhibiting neutrophil recruitment and activation.Front Pharmacol. 2022 Aug 8;13:949264. doi: 10.3389/fphar.2022.949264. eCollection 2022. Front Pharmacol. 2022. PMID: 36003515 Free PMC article.
References
-
- Alonso JM, Guiyoule A, et al. A model of meningococcal bacteremia after respiratory superinfection in influenza A virus-infected mice. FEMS Microbiol Lett. 2003;222(1):99–106. - PubMed
-
- Arimori Y, Nakamura R, et al. Type I interferon limits influenza virus-induced acute lung injury by regulation of excessive inflammation in mice. Antiviral Res. 2013;99(3):230–237. - PubMed
-
- Billiau A. Interferon: the pathways of discovery I. Molecular and cellular aspects. Cytokine Growth Factor Rev. 2006;17(5):381–409. - PubMed
-
- Bode JG, Ludwig S, et al. The MKK6/p38 mitogen-activated protein kinase pathway is capable of inducing SOCS3 gene expression and inhibits IL-6-induced transcription. Biol Chem. 2001;382(10):1447–1453. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

