Outer membrane lipoprotein biogenesis: Lol is not the end
- PMID: 26370942
- PMCID: PMC4632606
- DOI: 10.1098/rstb.2015.0030
Outer membrane lipoprotein biogenesis: Lol is not the end
Abstract
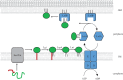
Bacterial lipoproteins are lipid-anchored proteins that contain acyl groups covalently attached to the N-terminal cysteine residue of the mature protein. Lipoproteins are synthesized in precursor form with an N-terminal signal sequence (SS) that targets translocation across the cytoplasmic or inner membrane (IM). Lipid modification and SS processing take place at the periplasmic face of the IM. Outer membrane (OM) lipoproteins take the localization of lipoproteins (Lol) export pathway, which ends with the insertion of the N-terminal lipid moiety into the inner leaflet of the OM. For many lipoproteins, the biogenesis pathway ends here. We provide examples of lipoproteins that adopt complex topologies in the OM that include transmembrane and surface-exposed domains. Biogenesis of such lipoproteins requires additional steps beyond the Lol pathway. In at least one case, lipoprotein sequences reach the cell surface by being threaded through the lumen of a beta-barrel protein in an assembly reaction that requires the heteropentomeric Bam complex. The inability to predict surface exposure reinforces the importance of experimental verification of lipoprotein topology and we will discuss some of the methods used to study OM protein topology.
Keywords: Bam complex; RcsF; localization of lipoproteins pathway; protein topology; surface-exposed protein.
© 2015 The Author(s).
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
