Host contributions to construction of three device-associated Candida albicans biofilms
- PMID: 26371129
- PMCID: PMC4645408
- DOI: 10.1128/IAI.00931-15
Host contributions to construction of three device-associated Candida albicans biofilms
Abstract
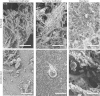
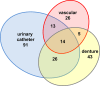
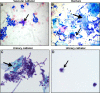
Among the most fascinating virulence attributes of Candida is the ability to transition to a biofilm lifestyle. As a biofilm, Candida cells adhere to a surface, such as a vascular catheter, and become encased in an extracellular matrix. During this mode of growth, Candida resists the normal immune response, often causing devastating disease. Based on scanning electron microscopy images, we hypothesized that host cells and proteins become incorporated into clinical biofilms. As a means to gain an understanding of these host-biofilm interactions, we explored biofilm-associated host components by using microscopy and liquid chromatography-mass spectrometry. Here we characterize the host proteins associated with several in vivo rat Candida albicans biofilms, including those from vascular catheter, denture, and urinary catheter models as well as uninfected devices. A conserved group of 14 host proteins were found to be more abundant during infection at each of the niches. The host proteins were leukocyte and erythrocyte associated and included proteins involved in inflammation, such as C-reactive protein, myeloperoxidase, and alarmin S100-A9. A group of 59 proteins were associated with both infected and uninfected devices, and these included matricellular and inflammatory proteins. In addition, site-specific proteins were identified, such as amylase in association with the denture device. Cellular analysis revealed neutrophils as the predominant leukocytes associating with biofilms. These experiments demonstrate that host cells and proteins are key components of in vivo Candida biofilms, likely with one subset associating with the device and another being recruited by the proliferating biofilm.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
Comparative Evaluations of the Pathogenesis of Candida auris Phenotypes and Candida albicans Using Clinically Relevant Murine Models of Infections.mSphere. 2020 Aug 5;5(4):e00760-20. doi: 10.1128/mSphere.00760-20. mSphere. 2020. PMID: 32759340 Free PMC article.
-
Fungal Biofilms: In Vivo Models for Discovery of Anti-Biofilm Drugs.Microbiol Spectr. 2015 Jun;3(3):E30. doi: 10.1128/microbiolspec.MB-0008-2014. Microbiol Spectr. 2015. PMID: 26397003 Free PMC article.
-
In vivo Candida glabrata biofilm development on foreign bodies in a rat subcutaneous model.J Antimicrob Chemother. 2015 Mar;70(3):846-56. doi: 10.1093/jac/dku447. Epub 2014 Nov 17. J Antimicrob Chemother. 2015. PMID: 25406296
-
Candida albicans Biofilms and Human Disease.Annu Rev Microbiol. 2015;69:71-92. doi: 10.1146/annurev-micro-091014-104330. Annu Rev Microbiol. 2015. PMID: 26488273 Free PMC article. Review.
-
Alternative Candida albicans lifestyles: growth on surfaces.Annu Rev Microbiol. 2005;59:113-33. doi: 10.1146/annurev.micro.59.030804.121034. Annu Rev Microbiol. 2005. PMID: 16153165 Review.
Cited by
-
Antifungal Compounds against Candida Infections from Traditional Chinese Medicine.Biomed Res Int. 2017;2017:4614183. doi: 10.1155/2017/4614183. Epub 2017 Dec 28. Biomed Res Int. 2017. PMID: 29445739 Free PMC article. Review.
-
Plasticity of Candida albicans Biofilms.Microbiol Mol Biol Rev. 2016 Jun 1;80(3):565-95. doi: 10.1128/MMBR.00068-15. Print 2016 Sep. Microbiol Mol Biol Rev. 2016. PMID: 27250770 Free PMC article. Review.
-
Vaginal Heparan Sulfate Linked to Neutrophil Dysfunction in the Acute Inflammatory Response Associated with Experimental Vulvovaginal Candidiasis.mBio. 2017 Mar 14;8(2):e00211-17. doi: 10.1128/mBio.00211-17. mBio. 2017. PMID: 28292981 Free PMC article.
-
How Biofilm Growth Affects Candida-Host Interactions.Front Microbiol. 2020 Jun 25;11:1437. doi: 10.3389/fmicb.2020.01437. eCollection 2020. Front Microbiol. 2020. PMID: 32670252 Free PMC article. Review.
-
Osteoarticular Mycoses.Clin Microbiol Rev. 2022 Dec 21;35(4):e0008619. doi: 10.1128/cmr.00086-19. Epub 2022 Nov 30. Clin Microbiol Rev. 2022. PMID: 36448782 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

