Counting tagged molecules one by one: Quantitative photoactivation and bleaching of photoactivatable fluorophores
- PMID: 26374025
- PMCID: PMC4567578
- DOI: 10.1063/1.4929991
Counting tagged molecules one by one: Quantitative photoactivation and bleaching of photoactivatable fluorophores
Abstract
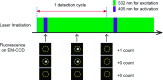
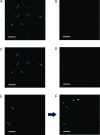
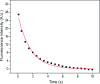
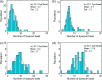
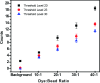
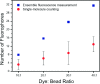
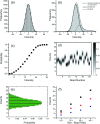
Determining the number of molecules in a given assembly, such as the number of proteins in a toxic aggregate, is often critical to understanding chemistry and function. Herein, we report a variation of a limitless method for counting photoactivatable fluorescent dyes in which single dye molecules are photoswitched to a fluorescent state, counted, and then irreversibly photobleached. We use this method to count the number of CAGE 552 covalently bound to the surface of 500 nm polystyrene beads. Activation of CAGE 552 was achieved with a 405 nm laser pulse. Once activated, the dye was excited with 532 nm light, and the fluorescence emission was collected with a CCD camera. The results from the fluorescence experiments were then compared to bulk fluorescence measurements to assess the error in counting. There are other ways of counting molecules, such as photobleaching and statistical analysis of reversible switchable chromophores. The method reported here provides a lower bound to the number of chromophores, with no upper limit to the number of molecules that can be quantified.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

