SPOP mutation leads to genomic instability in prostate cancer
- PMID: 26374986
- PMCID: PMC4621745
- DOI: 10.7554/eLife.09207
SPOP mutation leads to genomic instability in prostate cancer
Abstract
Genomic instability is a fundamental feature of human cancer often resulting from impaired genome maintenance. In prostate cancer, structural genomic rearrangements are a common mechanism driving tumorigenesis. However, somatic alterations predisposing to chromosomal rearrangements in prostate cancer remain largely undefined. Here, we show that SPOP, the most commonly mutated gene in primary prostate cancer modulates DNA double strand break (DSB) repair, and that SPOP mutation is associated with genomic instability. In vivo, SPOP mutation results in a transcriptional response consistent with BRCA1 inactivation resulting in impaired homology-directed repair (HDR) of DSB. Furthermore, we found that SPOP mutation sensitizes to DNA damaging therapeutic agents such as PARP inhibitors. These results implicate SPOP as a novel participant in DSB repair, suggest that SPOP mutation drives prostate tumorigenesis in part through genomic instability, and indicate that mutant SPOP may increase response to DNA-damaging therapeutics.
Keywords: DNA repair; SPOP; cancer genomics; cell biology; human; human biology; medicine; mouse; prostate cancer; zebrafish.
Conflict of interest statement
CEB: A patent (US Patent Application No: 2013/0331,279) has been issued to Weill Medical College of Cornell University on SPOP mutations in prostate cancer; is listed as co-inventor.
MAR: A patent (US Patent Application No: 2013/0331,279) has been issued to Weill Medical College of Cornell University on SPOP mutations in prostate cancer; is listed as co-inventor.
The other authors declare that no competing interests exist.
Figures

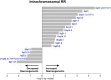


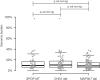
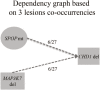


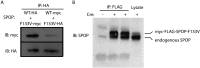


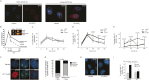

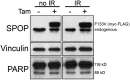
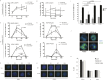
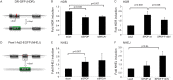



Comment in
-
SPOP the mutation.Elife. 2015 Oct 27;4:e11760. doi: 10.7554/eLife.11760. Elife. 2015. PMID: 26506153 Free PMC article.
References
-
- Audeh MW, Carmichael J, Penson RT, Friedlander M, Powell B, Bell-McGuinn KM, Scott C, Weitzel JN, Oaknin A, Loman N, Lu K, Schmutzler RK, Matulonis U, Wickens M, Tutt A. Oral poly (ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer: a proof-of-concept trial. Lancet. 2010;376:245–251. doi: 10.1016/S0140-6736(10)60893-8. - DOI - PubMed
-
- Baca SC, Prandi D, Lawrence MS, Mosquera JM, Romanel A, Drier Y, Park K, Kitabayashi N, MacDonald TY, Ghandi M, Van Allen E, Kryukov GV, Sboner A, Theurillat JP, Soong TD, Nickerson E, Auclair D, Tewari A, Beltran H, Onofrio RC, Boysen G, Guiducci C, Barbieri CE, Cibulskis K, Sivachenko A, Carter SL, Saksena G, Voet D, Ramos AH, Winckler W, Cipicchio M, Ardlie K, Kantoff PW, Berger MF, Gabriel SB, Golub TR, Meyerson M, Lander ES, Elemento O, Getz G, Demichelis F, Rubin MA, Garraway LA. Punctuated evolution of prostate cancer genomes. Cell. 2013;153:666–677. doi: 10.1016/j.cell.2013.03.021. - DOI - PMC - PubMed
-
- Barbieri CE, Baca SC, Lawrence MS, Demichelis F, Blattner M, Theurillat JP, White TA, Stojanov P, Van Allen E, Stransky N, Nickerson E, Chae SS, Boysen G, Auclair D, Onofrio RC, Park K, Kitabayashi N, MacDonald TY, Sheikh K, Vuong T, Guiducci C, Cibulskis K, Sivachenko A, Carter SL, Saksena G, Voet D, Hussain WM, Ramos AH, Winckler W, Redman MC, Ardlie K, Tewari AK, Mosquera JM, Rupp N, Wild PJ, Moch H, Morrissey C, Nelson PS, Kantoff PW, Gabriel SB, Golub TR, Meyerson M, Lander ES, Getz G, Rubin MA, Garraway LA. Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nature Genetics. 2012;44:685–689. doi: 10.1038/ng.2279. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

