Efficiency of CD19 chimeric antigen receptor-modified T cells for treatment of B cell malignancies in phase I clinical trials: a meta-analysis
- PMID: 26376680
- PMCID: PMC4741817
- DOI: 10.18632/oncotarget.5582
Efficiency of CD19 chimeric antigen receptor-modified T cells for treatment of B cell malignancies in phase I clinical trials: a meta-analysis
Abstract
Chimeric antigen receptor (CAR) modified T cells targeted CD19 showed promising clinical outcomes in treatment of B cell malignances such as chronic lymphocytic leukemia (CLL), acute lymphoblastic leukemia (ALL) and other indolent lymphomas. However, the clinical benefit varies tremendously among different trials. This meta-analysis investigated the efficacy (response rates and survival time) of CD19-CAR T cells in refractory B cell malignances in Phase I clinical trials. We searched publications between 1991 and 2014 from PubMed and Web of Science. Pooled response rates were calculated using random-effects models. Heterogeneity was investigated by subgroup analysis and meta-regression. Fourteen clinical trials including 119 patients were eligible for response rate evaluation, 62 patients in 12 clinical trials were eligible for progression-free survival analysis. The overall pooled response rate of CD19-CAR T cells was 73% (95% confidence interval [CI]: 46-94%). Significant heterogeneity across estimates of response rates was observed (p < 0.001, I2=88.3%). ALL patients have higher response rate (93%, 95% CI: 65-100%) than CLL (62%, 95% CI: 27-93%) and lymphoma patients (36%, 95% CI: 1-83%). Meta-regression analysis identified lymphodepletion and no IL-2 administrated T cells as two key factors associated with better clinical response. Lymphodepletion and higher infused CAR T cell number were associated with better prognosis. In conclusion, this meta-analysis showed a high clinical response rate of CD19-CAR T cell-based immunotherapy in treatment of refractory B cell malignancies. Lymphodepletion and increasing number of infused CD19-CAR T cells have positive correlations with the clinical efficiency, on the contrary, IL-2 administration to T cells is not recommended.
Keywords: B cell malignancies; CD19; chimeric antigen receptor; efficiency; meta analysis.
Conflict of interest statement
The authors have declared that no conflict of interest exists.
Figures

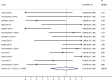


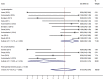

Similar articles
-
The efficacy of anti-CD19 chimeric antigen receptor T cells for B-cell malignancies.Cytotherapy. 2019 Jul;21(7):769-781. doi: 10.1016/j.jcyt.2019.04.005. Epub 2019 May 31. Cytotherapy. 2019. PMID: 31160157
-
The severe cytokine release syndrome in phase I trials of CD19-CAR-T cell therapy: a systematic review.Ann Hematol. 2018 Aug;97(8):1327-1335. doi: 10.1007/s00277-018-3368-8. Epub 2018 May 15. Ann Hematol. 2018. PMID: 29766234
-
Factors associated with outcomes after a second CD19-targeted CAR T-cell infusion for refractory B-cell malignancies.Blood. 2021 Jan 21;137(3):323-335. doi: 10.1182/blood.2020006770. Blood. 2021. PMID: 32967009 Free PMC article. Clinical Trial.
-
Durable Molecular Remissions in Chronic Lymphocytic Leukemia Treated With CD19-Specific Chimeric Antigen Receptor-Modified T Cells After Failure of Ibrutinib.J Clin Oncol. 2017 Sep 10;35(26):3010-3020. doi: 10.1200/JCO.2017.72.8519. Epub 2017 Jul 17. J Clin Oncol. 2017. PMID: 28715249 Free PMC article. Clinical Trial.
-
CAR T Cell Therapy in Acute Lymphoblastic Leukemia and Potential for Chronic Lymphocytic Leukemia.Curr Treat Options Oncol. 2016 Jun;17(6):28. doi: 10.1007/s11864-016-0406-4. Curr Treat Options Oncol. 2016. PMID: 27098534 Review.
Cited by
-
Cardiovascular effects associated with chimeric antigen receptor T cell therapy in cancer patients: A meta-analysis.Front Oncol. 2022 Nov 9;12:924208. doi: 10.3389/fonc.2022.924208. eCollection 2022. Front Oncol. 2022. PMID: 36439485 Free PMC article.
-
Efficiency and safety of autologous chimeric antigen receptor T-cells therapy used for patients with lymphoma: A systematic review and meta-analysis.Medicine (Baltimore). 2019 Oct;98(42):e17506. doi: 10.1097/MD.0000000000017506. Medicine (Baltimore). 2019. PMID: 31626107 Free PMC article.
-
Tissue factor (coagulation factor III): a potential double-edge molecule to be targeted and re-targeted toward cancer.Biomark Res. 2023 Jun 6;11(1):60. doi: 10.1186/s40364-023-00504-6. Biomark Res. 2023. PMID: 37280670 Free PMC article. Review.
-
T-cell therapies for HIV: Preclinical successes and current clinical strategies.Cytotherapy. 2016 Aug;18(8):931-942. doi: 10.1016/j.jcyt.2016.04.007. Epub 2016 Jun 2. Cytotherapy. 2016. PMID: 27265874 Free PMC article. Review.
-
CD19 chimeric antigen receptor (CD19 CAR)-redirected adoptive T-cell immunotherapy for the treatment of relapsed or refractory B-cell Non-Hodgkin's Lymphomas.Am J Cancer Res. 2016 Jan 15;6(2):403-24. eCollection 2016. Am J Cancer Res. 2016. PMID: 27186412 Free PMC article. Review.
References
-
- Kochenderfer JN, Dudley ME, Carpenter RO, Kassim SH, Rose JJ, Telford WG, Hakim FT, Halverson DC, Fowler DH, Hardy NM, Mato AR, Hickstein DD, Gea-Banacloche JC, et al. Donor-derived CD19-targeted T cells cause regression of malignancy persisting after allogeneic hematopoietic stem cell transplantation. Blood. 2013;122:4129–39. - PMC - PubMed
-
- Cruz CR, Micklethwaite KP, Savoldo B, Ramos CA, Lam S, Ku S, Diouf O, Liu E, Barrett AJ, Ito S, Shpall EJ, Krance RA, Kamble RT, et al. Infusion of donor-derived CD19-redirected virus-specific T cells for B-cell malignancies relapsed after allogeneic stem cell transplant: a phase 1 study. Blood. 2013;122:2965–73. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

