An in silico Approach Reveals Associations between Genetic and Epigenetic Factors within Regulatory Elements in B Cells from Primary Sjögren's Syndrome Patients
- PMID: 26379672
- PMCID: PMC4549647
- DOI: 10.3389/fimmu.2015.00437
An in silico Approach Reveals Associations between Genetic and Epigenetic Factors within Regulatory Elements in B Cells from Primary Sjögren's Syndrome Patients
Abstract
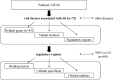
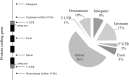
Recent advances in genetics have highlighted several regions and candidate genes associated with primary Sjögren's syndrome (SS), a systemic autoimmune epithelitis that combines exocrine gland dysfunctions, and focal lymphocytic infiltrations. In addition to genetic factors, it is now clear that epigenetic deregulations are present during SS and restricted to specific cell type subsets, such as lymphocytes and salivary gland epithelial cells. In this study, 72 single nucleotide polymorphisms (SNPs) associated with 43 SS gene risk factors were selected from publicly available and peer reviewed literature for further in silico analysis. SS risk variant location was tested revealing a broad distribution in coding sequences (5.6%), intronic sequences (55.6%), upstream/downstream genic regions (30.5%), and intergenic regions (8.3%). Moreover, a significant enrichment of regulatory motifs (promoter, enhancer, insulator, DNAse peak, and expression quantitative trait loci) characterizes SS risk variants (94.4%). Next, screening SNPs in high linkage disequilibrium (r (2) ≥ 0.8 in Caucasians) revealed 645 new variants including 5 SNPs with missense mutations, and indicated an enrichment of transcriptionally active motifs according to the cell type (B cells > monocytes > T cells ≫ A549). Finally, we looked at SS risk variants for histone markers in B cells (GM12878), monocytes (CD14(+)) and epithelial cells (A548). Active histone markers were associated with SS risk variants at both promoters and enhancers in B cells, and within enhancers in monocytes. In conclusion and based on the obtained in silico results that need further confirmation, associations were observed between SS genetic risk factors and epigenetic factors and these associations predominate in B cells, such as those observed at the FAM167A-BLK locus.
Keywords: B cells; Sjögren’s syndrome; epigenetics; genetics; histone modifications; in silico; polymorphism.
Figures


References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

