Managing chronic myeloid leukaemia in the elderly with intermittent imatinib treatment
- PMID: 26383820
- PMCID: PMC4648524
- DOI: 10.1038/bcj.2015.75
Managing chronic myeloid leukaemia in the elderly with intermittent imatinib treatment
Abstract
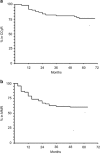
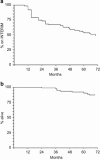
The aim of this study was to investigate the effects of a non-standard, intermittent imatinib treatment in elderly patients with Philadelphia-positive chronic myeloid leukaemia and to answer the question on which dose should be used once a stable optimal response has been achieved. Seventy-six patients aged ⩾65 years in optimal and stable response with ⩾2 years of standard imatinib treatment were enrolled in a study testing a regimen of intermittent imatinib (INTERIM; 1-month on and 1-month off). With a minimum follow-up of 6 years, 16/76 patients (21%) have lost complete cytogenetic response (CCyR) and major molecular response (MMR), and 16 patients (21%) have lost MMR only. All these patients were given imatinib again, the same dose, on the standard schedule and achieved again CCyR and MMR or an even deeper molecular response. The probability of remaining on INTERIM at 6 years was 48% (95% confidence interval 35-59%). Nine patients died in remission. No progressions were recorded. Side effects of continuous treatment were reduced by 50%. In optimal and stable responders, a policy of intermittent imatinib treatment is feasible, is successful in about 50% of patients and is safe, as all the patients who relapsed could be brought back to optimal response.
Conflict of interest statement
Domenico Russo, Patrizia Pregno and Simona Soverini receives compensation as a consultant for Novartis, Bristol-Myers Squibb and Ariad. Elisabetta Abruzzese receives compensation as a consultant for Ariad, Bristol-Myers Squibb, Novartis, Takeda and Pfizer. Giovanni Martinelli receives compensation as a consultant for Novartis, Bristol-Myers Squibb and Pfizer. Gianantonio Rosti, Fausto Castagnetti and Michele Baccarani receives compensation as a consultant for Novartis, Ariad, Bristol-Myers Squibb and Pfizer. Mario Tiribelli receives compensation as a consultant for Novartis, BMS and Ariad. The remaining authors declare no conflict of interest.
Figures

References
-
- 1Hehlmann R, Hochhaus A, Baccarani M. Chronic myeloid leukaemia. Lancet 2005; 370: 342–350. - PubMed
-
- 5National Comprehensive Cancer Network: NCCN. Clinical Practice Guidelines in Oncology: Chronic Myeloid Leukemia, version 1, 2015. www.nccn.org/professionals/physician_gls/pdf/cml.pdf. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

