Recommendations and evidence for reporting items in pediatric clinical trial protocols and reports: two systematic reviews
- PMID: 26385379
- PMCID: PMC4574457
- DOI: 10.1186/s13063-015-0954-0
Recommendations and evidence for reporting items in pediatric clinical trial protocols and reports: two systematic reviews
Abstract
Background: Complete and transparent reporting of clinical trial protocols and reports ensures that these documents are useful to all stakeholders, that bias is minimized, and that the research is not wasted. However, current studies repeatedly conclude that pediatric trial protocols and reports are not appropriately reported. Guidelines like SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) and CONSORT (Consolidated Standards of Reporting Trials) may improve reporting, but do not offer guidance on issues unique to pediatric trials. This paper reports two systematic reviews conducted to build the evidence base for the development of pediatric reporting guideline extensions: 1) SPIRIT-Children (SPIRIT-C) for pediatric trial protocols, and 2) CONSORT-Children (CONSORT-C) for pediatric trial reports.
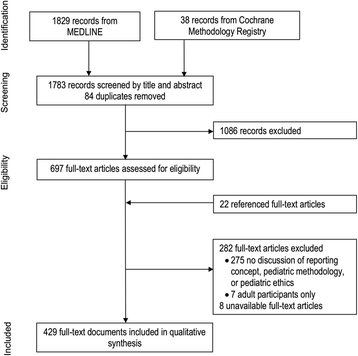
Method: MEDLINE, the Cochrane Methodology Register, and reference lists of included studies were searched. Publications of any type were eligible if they included explicit recommendations or empirical evidence for the reporting of potential items in a pediatric protocol (SPIRIT-C systematic review) or trial report (CONSORT-C systematic review). Study characteristics, recommendations and evidence for pediatric extension items were extracted. Recurrent themes in the recommendations and evidence were identified and synthesized. All steps were conducted by two reviewers.
Results: For the SPIRIT-C and CONSORT-C systematic reviews 366 and 429 publications were included, respectively. Recommendations were identified for 48 of 50 original reporting items and sub-items from SPIRIT, 15 of 20 potential SPIRIT-C reporting items, all 37 original CONSORT items and sub-items, and 16 of 22 potential CONSORT-C reporting items. The following overarching themes of evidence to support or refute the utility of reporting items were identified: transparency; reproducibility; interpretability; usefulness; internal validity; external validity; reporting bias; publication bias; accountability; scientific soundness; and research ethics.
Conclusion: These systematic reviews are the first to systematically gather evidence and recommendations for the reporting of specific items in pediatric protocols and trials. They provide useful and translatable evidence on which to build pediatric extensions to the SPIRIT and CONSORT reporting guidelines. The resulting SPIRIT-C and CONSORT-C will provide guidance to the authors of pediatric protocols and reports, respectively, helping to alleviate concerns of inappropriate and inconsistent reporting, and reduce research waste.
Figures
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical