Specific biological responses of the synovial membrane to carbon nanotubes
- PMID: 26388047
- PMCID: PMC4585697
- DOI: 10.1038/srep14314
Specific biological responses of the synovial membrane to carbon nanotubes
Abstract
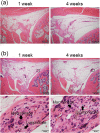
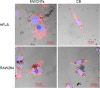
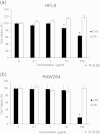
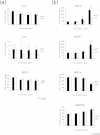
Biological evaluation of carbon nanotubes (CNTs) is typically performed in the lung or abdominal cavity; however, biological reactions to CNTs are predicted to be markedly different in other tissues. In applications of CNTs as reinforcement for artificial joints and drug delivery systems, including their use in bone regeneration, the intra-articular synovial membrane makes contact with the CNTs. Herein, we analyzed the reaction of the synovial membrane with multiwalled CNTs (MWCNTs). Injection of MWCNTs into rat knee joints revealed their dose-dependent incorporation into deep synovial membranes and the formation of granulation tissue, without long-term inflammation. MWCNTs were incorporated into human fibroblast-like synoviocytes (HFLSs), with less cytotoxicity than that observed in macrophages (RAW264 cells). Moreover, MWCNTs inhibited the release of cytokines and chemokines from HFLSs. The reaction of the synovial membrane with MWCNTs differed from that observed in other tissues; thus, detailed biological evaluation at each target site is necessary for clinical applications.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

Similar articles
-
Carbon nanotubes stimulate synovial inflammation by inducing systemic pro-inflammatory cytokines.Nanoscale. 2016 Oct 27;8(42):18070-18086. doi: 10.1039/c6nr06041b. Nanoscale. 2016. PMID: 27714147
-
Multiwalled carbon nanotubes induce a fibrogenic response by stimulating reactive oxygen species production, activating NF-κB signaling, and promoting fibroblast-to-myofibroblast transformation.Chem Res Toxicol. 2011 Dec 19;24(12):2237-48. doi: 10.1021/tx200351d. Epub 2011 Nov 22. Chem Res Toxicol. 2011. PMID: 22081859
-
Suppression of human arthritis synovial fibroblasts inflammation using dexamethasone-carbon nanotubes via increasing caveolin-dependent endocytosis and recovering mitochondrial membrane potential.Int J Nanomedicine. 2017 Aug 10;12:5761-5779. doi: 10.2147/IJN.S142122. eCollection 2017. Int J Nanomedicine. 2017. PMID: 28848352 Free PMC article.
-
A review on protein functionalized carbon nanotubes.J Appl Biomater Funct Mater. 2015 Dec 18;13(4):e301-12. doi: 10.5301/jabfm.5000231. J Appl Biomater Funct Mater. 2015. PMID: 26660626 Review.
-
Rheumatoid arthritis: targeting the proliferative fibroblasts.Prog Cell Cycle Res. 2003;5:59-70. Prog Cell Cycle Res. 2003. PMID: 14593701 Review.
Cited by
-
Future Prospects for Clinical Applications of Nanocarbons Focusing on Carbon Nanotubes.Adv Sci (Weinh). 2022 Aug;9(24):e2201214. doi: 10.1002/advs.202201214. Epub 2022 Jun 26. Adv Sci (Weinh). 2022. PMID: 35754236 Free PMC article.
-
A three-dimensional block structure consisting exclusively of carbon nanotubes serving as bone regeneration scaffold and as bone defect filler.PLoS One. 2017 Feb 24;12(2):e0172601. doi: 10.1371/journal.pone.0172601. eCollection 2017. PLoS One. 2017. PMID: 28235026 Free PMC article.
-
Biokinetic Evaluation of Contrast Media Loaded Carbon Nanotubes Using a Radiographic Device.Toxics. 2021 Dec 2;9(12):331. doi: 10.3390/toxics9120331. Toxics. 2021. PMID: 34941765 Free PMC article.
-
Intra-articular targeting of nanomaterials for the treatment of osteoarthritis.Acta Biomater. 2019 Jul 15;93:239-257. doi: 10.1016/j.actbio.2019.03.010. Epub 2019 Mar 9. Acta Biomater. 2019. PMID: 30862551 Free PMC article. Review.
-
Knee Osteoarthritis Therapy: Recent Advances in Intra-Articular Drug Delivery Systems.Drug Des Devel Ther. 2022 May 4;16:1311-1347. doi: 10.2147/DDDT.S357386. eCollection 2022. Drug Des Devel Ther. 2022. PMID: 35547865 Free PMC article. Review.
References
-
- Takagi A. et al. Induction of mesothelioma in p53+/− mouse by intraperitoneal application of multi-wall carbon nanotube. J. Toxicol. Sci. 33, 105–116 (2008). - PubMed
-
- Cavallo D. et al. Multi-walled carbon nanotubes induce cytotoxicity and genotoxicity in human lung epithelial cells. J. Appl. Toxicol. 32, 454–464 (2012). - PubMed
-
- Li R. et al. In vivo detection of magnetic labeled oxidized multi-walled carbon nanotubes by magnetic resonance imaging. Nanotechnology 25, 495102 (2014). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

