Design, Immune Responses and Anti-Tumor Potential of an HPV16 E6E7 Multi-Epitope Vaccine
- PMID: 26390407
- PMCID: PMC4577214
- DOI: 10.1371/journal.pone.0138686
Design, Immune Responses and Anti-Tumor Potential of an HPV16 E6E7 Multi-Epitope Vaccine
Abstract
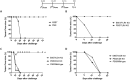
Cervical cancer is a common type of cancer among women worldwide and infection with high-risk human papillomavirus (HPVs) types represents the major risk factor for the etiopathogenesis of the disease. HPV-16 is the most frequently identified HPV type in cervical lesions and expression of E6 and E7 oncoproteins is required for the uncontrolled cellular proliferation. In the present study we report the design and experimental testing of a recombinant multi-epitope protein containing immunogenic epitopes of HPV-16 E6 and E7. Tumor preventive assays, based on the engraftment of TC-1 cells in mice, showed that the E6E7 multi-epitope protein induced a full preventive anti-tumor protection in wild-type mice, as well as in mice deficient in expression of CD4+ T cells and TLR4 receptor. Nonetheless, no anti-tumor protection was observed in mice deficient in CD8+ T cells. Also, the vaccine promoted high activation of E6/E7-specific T cells and in a therapeutic-approach, E6E7 protein conferred full anti-tumor protection in mice. These results show a potential use of this E6E7 multi-epitope antigen as a new and promising antigen for the development of a therapeutic vaccine against tumors induced by HPV.
Conflict of interest statement
Figures


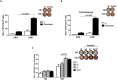
References
-
- Liu TY, Hussein WM, Toth I, Skwarczynski M. Advances in peptide-based human papillomavirus therapeutic vaccines. Current topics in medicinal chemistry. 2012; 12: 1581–92. - PubMed
-
- Robinson W III. Management of cervical neoplasia. Cancer treatment and research. 2001; 104: 287–302. - PubMed
-
- Diniz MO, Lasaro MO, Ertl HC, Ferreira LC. Immune responses and therapeutic antitumor effects of an experimental DNA vaccine encoding human papillomavirus type 16 oncoproteins genetically fused to herpesvirus glycoprotein D. Clinical and vaccine immunology: CVI. 2010; 17: 1576–83. 10.1128/CVI.00264-10 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

